March 2005 Issue Table of Contents
Annealing of gem-quality diamonds at very high pressures (above 5 GPa) and temperatures (above ~1800°C) can produce significant changes in their color. Treatment under these high-pressure–hightemperature (HPHT) conditions affects certain optically active defects and their absorptions in the visible spectrum. In the jewelry industry, laboratorytreated diamonds are valued much less than those of natural color. Polished diamonds are carefully examined at gemological laboratories to determine the “origin of color” as part of an overall assessment of their quality. Currently, the recognition of HPHT-treated diamonds involves the determination of various visual properties (such as color and features seen under magnification), as well as characterization by several spectroscopic techniques. HPHT-treated diamonds were introduced into the jewelry trade in the late 1990s, and despite progress in their recognition, their identification remains a challenge. While some detection methodologies have been established, the large number of diamonds requiring testing with sophisticated analytical instrumentation poses a logistical problem for gemological laboratories.
KEYWORDS: Diamond, gemstone, color, treatment, HPHT, annealing, identification
INTRODUCTION
Diamonds are the most important gemstones, and their sales are the foundation of the international jewelry industry. Gem diamonds hold a special fascination, as evidenced by the popularity of, and media interest in, exhibitions and events featuring them (e.g., King and Shigley 2003). In 2003, retail diamond jewelry sales in the United States amounted to over US$29 billion. Continued confidence in the jewelry marketplace depends both on an accurate determination of whether a diamond (or any gemstone) is natural, synthetic, or laboratory-treated and on a full disclosure of this information at the time of sale.
Commercial treatments to improve the color of gem diamonds have existed since the 1930s (see Nassau 1994, pp. 141–151). These processes mostly involve exposure to highenergy radiation, sometimes followed by heating to temperatures up to several hundred degrees Celsius or more. Over time, diamond color (as well as clarity) treatments have become both more sophisticated and widespread, and new treatment processes have been developed. In some cases, a gemologist, using just a binocular microscope and a simple spectroscope, can recognize color-treated diamonds. However, detecting them often requires more sophisticated instruments such as UV–visible or photoluminescence spectrometers. For some treated diamonds, this determination still cannot be made with certainty; this is especially true when the treatment processes (e.g., irradiation) mimic natural processes that affect diamonds in the Earth. The fact that some natural-color gem diamonds sell at prices in excess of US$100,000 or more per carat emphasizes the importance of a correct “origin of color” determination.
This article deals with the most difficult current identification challenge—the recognition of diamonds that have had their color either removed or changed by annealing at high pressures and temperatures (referred to as HPHT treatment).
DIAMOND TYPES AND COLORATION
Diamonds can be divided into categories, called types, based on differences in certain physical properties, particularly optical absorption. Initially, diamonds with strong absorptions of both IR and UV radiation were designated as Type I, whereas those without were labeled Type II (Robertson et al. 1934). These “types” are related to the presence of impurities (see TABLE 1). The boundaries between categories are somewhat arbitrary, and mixed types are possible within a single diamond.
The structural arrangements of these impurities within the diamond lattice are perhaps more interesting than their abundances. Nitrogen (and to a lesser extent boron and hydrogen), along with point and extended defects, cause absorptions in the visible spectrum, which give rise to coloration (see Table 1).
Certain colors in diamond are due not to trace-element impurities, but to absorptions resulting from various optically active defects. In brown and pink diamonds, the “color centers” (which produce a broad region of absorption centered at about 550 nm) are thought to be caused by dislocations and point defects that result from deformation of the diamonds during their extended storage in the Earth. Exposure of diamonds to radiation, either in nature or the laboratory, creates defects that cause green and some blue colors (due to the GR center at 741 nm and its associated sidebands, see Table 1; for further information, see Fritsch 1998; Collins 2001).
HPHT-TREATMENT OF DIAMONDS
Experimental studies in the 1970s showed that the degree of aggregation of nitrogen atoms could be increased or decreased by heating Type I diamonds to very high temperatures at high pressures (see Evans and Rainey 1975; Brozel et al. 1978). This was accompanied by either a weakening or an intensification of the yellow color. Because of the difficulty in achieving these conditions, this procedure was not commercially viable as a treatment of gem diamonds.
In March 1999, the jewelry industry was taken by surprise when the prominent U.S. jewelry firm, Lazare Kaplan International, announced a new process developed by the General Electric (GE) Company for transforming certain Type IIa diamonds from brown to colorless (see p. 101). Initially sold under the name “GE POL” (for Pegasus Overseas Ltd., a foreign GE subsidiary), these color-treated diamonds are currently marketed under the trade name “Bellataire” (e.g., http://www.ge.com/uk/bellataire/). No details of the process were provided by GE at the time (patents have since been published; see Vagarali et al. 2004). However, it was subsequently established that the technique involved annealing diamonds for brief periods of time at very high temperatures and pressures (1800 to ~2700°C, and at 5 to 9 GPa to prevent transformation to graphite). Under such conditions, lattice-scale defects in the structure that produce brown coloration are annealed out, thereby rendering the diamonds colorless.
This process is carried out with the same equipment typically used for diamond synthesis, including the “belt”, tetrahedral, cubic, and octahedral presses, as well as the Russian-designed “BARS” units (= bespressovye apparaty tipa razreznaya sfera, or “split sphere no press apparatus”). One or more rough or polished diamonds are loaded into a capsule, which is then placed in the apparatus. The exact P and T used depend on the type of diamonds being treated, the color change desired, and the equipment configuration, with annealing times being as short as a few minutes. At the end of a treatment run, the diamonds may have a black, graphitized outer surface, which can be removed by heating to several hundred degrees Celsius for a short time in a laboratory oven. Following treatment, all diamonds exhibit surface damage in the form of etching and pitting, so after processing, a cut diamond requires repolishing. Polished diamonds of all sizes (0.01 to more than 30 ct) have been treated by the HPHT process.
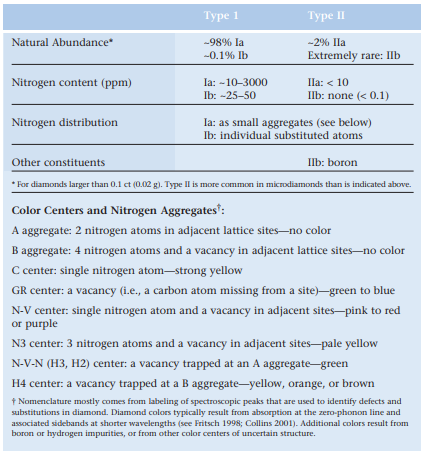
Annealing of Type IIa brown diamonds under HPHT conditions removes structural defects by means of plastic flow (Schmetzer 1999; Collins et al. 2000). This decreases the broad absorption in the visible spectrum that causes the brown coloration (Fisher and Spits 2000; also see FIGS. 1 AND 2). It is also possible to change certain very rare brown-pink and grayblue Type II diamonds by eliminating the brown component of their coloration and thereby enhancing the underlying pink or blue color, respectively (Hall and Moses 2000, 2001). Wang et al. (2003) recently described an intensely colored green-yellow Type IIa diamond that had been treated by this method.


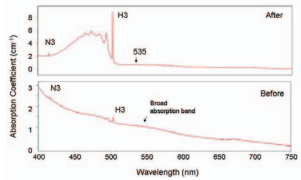
Initially this treatment focused on removal of brown color from Type IIa diamonds, but experiments on changing the color of Type I diamonds soon followed (De Weerdt and Van Royen 2000; Reinitz et al. 2000; De Weerdt and Collins 2003). Brownish-yellow Type Ia diamonds can be transformed to yellow, orange-yellow, or green-yellow as annealing permits vacancies to migrate and combine with aggregated nitrogen to form nitrogen-vacancy-nitrogen (N-V-N) centers that give rise to strong green coloration (due to the H3 optical center; Collins et al. 2000; see FIG. 3). At higher T, some A nitrogen centers are also broken up, forming C centers. This is accompanied by H3 changing to H2 centers and the formation of the nitrogen-vacancy (N-V) centers (see Collins 2001; Zaitsev 2001). Only recently have Type 1a diamonds been reported whose light yellow color is the result of HPHT treatment.
When the challenge for gemological laboratories focused on detecting decolorized Type IIa diamonds, the problem was limited because of their rarity. Now, with the treatment of Type I diamonds added, the identification problem is much greater because the latter are far more abundant and more variable in gemological properties.
DETECTING HPHT-ANNEALED DIAMONDS
HPHT treatment presented significant challenges for the jewelry industry in terms of identification and disclosure. Even though only a relatively small number were initially involved, HPHT-treated diamonds often exhibited few if any visual clues for a jeweler or gemologist to detect this process. As an aid to recognition, General Electric inscribed an identification mark easily visible at 10× magnification on the polished “girdle” surface of each of their treated diamonds (those sold by other companies may or may not have similar laser inscriptions). However, these marks can be removed by repolishing the inscribed surface.
HPHT-treated diamonds can exhibit visual features that are lacking in untreated diamonds, for example, the above-mentioned surface damage. They can also display graphitized fracture surfaces and inclusions (see Chalain et al. 1999; Moses et al. 1999). Colorless Type IIa diamonds display greater transparency to short-wave (265 nm) UV radiation than the more abundant Type Ia diamonds, and this offers a preliminary way to separate the former for further testing (Moses et al. 1999; Chalain et al. 2000).
Since visual features are not always present in polished diamonds, detection of this treatment requires the use of spectroscopic techniques (Collins 2001). For example, in HPHTtreated Type II diamonds, Chalain et al. (1999, 2000) interpreted the 637 nm absorption peak (due to the neutral N-V center) as a potential distinctive feature (also see Fritsch et al. 2001). Fisher and Spits (2000) described the broad 270 nm band (due to C nitrogen centers) seen in the spectra of HPHT-treated diamonds with increasing yellow coloration. Using 514-nm Ar-laser excitation, they also reported that the photoluminescence (PL) peaks at 575 and 637 nm (due to neutral and negative charge states at N-V centers, respectively) have an intensity ratio (637>575) in treated diamonds that differs from that in untreated Type IIa diamonds (575>637). Other non-destructive analytical tools, including X-ray topography and cathodoluminescence, have been tried, but further work is needed on a larger population of untreated and HPHT-treated diamonds to support their use for detection purposes (e.g., Smith et al. 2000).
In yellow Type Ia diamonds, HPHT treatment produces new absorption features while again reducing the brown coloration (Van Royen and Pal’yanov 2002). For example, strong green H3 luminescence, along with the contribution of the sideband of the H2 center, causes the treated diamonds to be yellow-green to green (Collins et al. 2000; Collins 2001; see Fig. 4). Just as in Type IIa diamonds, isolated nitrogen (C centers) are formed in Type Ia diamonds by annealing, as can be seen in mid-infrared spectra (Reinitz et al. 2000; De Weerdt and Van Royen 2000). Current gemological research is focused on confirming the use of spectral features in both diamond types and on establishing additional means of treated-diamond identification.
Several potential detection techniques have been developed to quickly check diamonds to see if they have been HPHT treated. A patent issued to General Electric describes a method using the presence or absence of one or more PL lines for this purpose (Anthony et al. 2002). Researchers at the De Beers Diamond Trading Company (DTC) Research Centre have developed a prototype screening instrument that again is based on the detection of spectral features (Welbourn and Williams 2002). At gemological laboratories, separation of natural-color from HPHT-treated diamonds is based on as many features as possible. Development of identification criteria rests on assembling a database of information on large numbers of known untreated and known treated diamonds. Creation of such a database represents a significant research activity in the major diamond-testing gemological laboratories.
CONCLUSION
Gemologists face continued challenges in recognizing natural, treated, and synthetic gem materials. HPHT-treated diamonds pose a special difficulty since some colors produced correspond to those of high-priced untreated natural diamonds, and yet they are valued very differently. Their detection often requires the use of sophisticated scientific equipment. Treatment experiments undertaken by the Gemological Institute of America and other researchers have been able to reproduce the kinds of color changes in diamonds that can be brought about by HPHT annealing. Systematic documentation of the gemological properties of a vast number of treated and untreated diamonds has yielded new identification criteria. Nonetheless, the distinction of HPHT-annealed diamonds represents a difficult challenge for gem-testing laboratories that receive numerous gems on a daily basis. Despite much progress, the detection of HPHTtreated diamonds will remain a concern for the jewelry industry as the use of this process becomes more widespread.
REFERENCES
Anthony TR, Casey JK, Smith AC, Vagarali SS (2002) Method of detection of natural diamonds that have been processed at high pressure and high temperatures. U.S. Patent 6,377,340 B1, dated 23 April 2002
Brozel MR, Evans T, Stephenson RF (1978) Partial dissociation of nitrogen aggregates in diamond by high-temperature high-pressure treatments. Proceedings Royal Society of London A361: 109-127
Chalain J-P, Fritsch E, Hänni H (1999) Détection des diamants GE POL: une première étape. Revue de Gemmologie a.f.g. 138/139: 2-11
Chalain J-P, Fritsch E, Hänni H (2000) Identification of GE POL diamonds: a second step. Journal of Gemmology 27: 73-78
Collins AT (2001) The colour of diamond and how it may be changed. Journal of Gemmology 27: 341-359
Collins AT, Kanda H, Kitawaki H (2000) Colour changes produced in natural brown diamonds by high-pressure, high-temperature treatment. Diamond and Related Materials 9: 113-122
De Weerdt F, Van Royen J (2000) Investigation of seven diamonds, HPHT treated by NovaDiamond. Journal of Gemmology 27: 201-208
De Weerdt F, Collins AT (2003) The influence of pressure on high-pressure, high-temperature annealing of type Ia diamond. Diamond and Related Materials 12: 507-510
Evans T, Rainey P (1975) Changes in the defect structure of diamond due to high temperature–high pressure treatment. Diamond Research 1975: 29–34
Fisher D, Spits RA (2000) Spectroscopic evidence of GE POL HPHT-treated natural type IIa diamonds. Gems & Gemology 36: 42-49
Fritsch E (1998) The nature of color in diamonds. In: Harlow GE (ed) The nature of diamonds, Cambridge University Press, Cambridge, pp 23-77
Fritsch E, Chalain J-P, Hänni H (2001) Identification of GE POLTM diamonds. Australian Gemmologist 21: 172-177
Hall M, Moses TM (2000) Gem Trade Lab Notes: Diamond – blue and pink, HPHT annealed. Gems & Gemology 36: 254-255
Hall M, Moses TM (2001) Gem Trade Lab Notes: Update on blue and pink HPHT-annealed diamonds. Gems & Gemology 37: 215-216
King JM, Shigley JE (2003) An important exhibit of seven rare gem diamonds. Gems & Gemology 39: 136-143
Moses TM, Shigley JE, McClure SF, Koivula JI, van Daele M (1999) Observations on GE-processed diamonds: A photographic record. Gems & Gemology 35: 14-22
Nassau K (1994) Gemstone enhancement – history, science and state of the art, 2nd Edition. Butterworth-Heinemann Ltd, Oxford, 252 pp
Reinitz I, Buerki PR, Shigley JE, McClure SF, Moses TM (2000) Identification of HPHT-treated yellow to green diamonds. Gems & Gemology 36: 128-137
Robertson R, Fox JJ, Martin AE (1934) Two types of diamond. Philosophical Transactions of the Royal Society of London A232: 463-535
Schmetzer K (1999) Clues to the process used by General Electric to enhance the GE POL diamonds. Gems & Gemology 35: 186-190
Smith CP, Bosshart G, Ponahlo J, Hammer VMF, Klapper H, Schmetzer K (2000) GE POL diamonds: Before and after. Gems & Gemology 36: 192-215
Vagarali SS, Webb SW, Jackson WE, Banholzer WF, Anthony TR, Kaplan GR (2004) High pressure/high temperature production of colorless and fancy-colored diamonds. U.S. Patent 6,692,714, dated 17 February 2004
Van Royen J, Pal’yanov YN (2002) High- pressure–high-temperature treatment of natural diamonds. Journal of Physics – Condensed Matter 14: 10953-10956
Wang W, Hall M, Moses TMM (2003) Gem Trade Lab Notes: Diamond – Intensely colored type IIa with substantial nitrogen-related defects. Gems & Gemology 39: 39-41
Welbourn C, Williams R (2002) DTC Research comes to diamond’s defence. Rapaport Diamond Report 25 (3 May): 44-45
Zaitsev AM (2001) Optical properties of diamond – a data handbook. Springer- Verlag, Berlin, 502 pp

