Soil Biogeochemical Processes within the Critical Zone
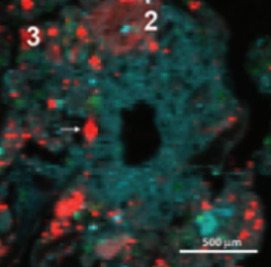
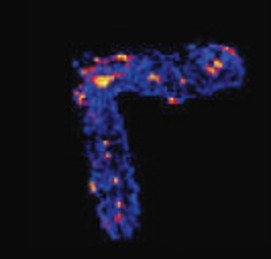
Many processes that affect soil and water quality occur at the water wetted interface of weathering products such as clays, oxides, and organic matter. Especially near the sunlit surface of the Critical Zone, these interfaces associate with plant roots and soil organisms to form porous, aggregated structures. Soil aggregates and intervening pore networks give rise to a patchwork of interconnected microenvironments. The ensuing steep geochemical gradients affect weathering processes, fuel the activities of microbes, and drive interfacial reactions that retain and transform rock- or ecosystem-derived chemicals and anthropogenic pollutants.
Soil Biogeochemical Processes within the Critical Zone Read More »