August 2025 Issue Table of Contents
Re-Os geochronology is a powerful tool for unravelling the complexity of petroleum systems. Because of their organophile nature, Re and Os are enriched in sedimentary organic matter and the hydrocarbons it produces upon heating. Rhenium and Os isotopes are used to date hydrocarbon generation, to fingerprint oils, to distinguish different pulses of oil generation, and to determine interactions between oil, host rock, and formation water. Here we summarize knowledge on the content, distribution, and isotopic composition of Re and Os in petroleum and associated waters, highlight the main technical advances for Re-Os analyses, review experimental studies on water–oil interaction, and discuss the diverse Re-Os applications to petroleum systems.
1811-5209/25/0021-0271$2.50 DOI: 10.2138/gselements.21.4.271
Keywords: Petroleum systems; oil; bitumen; kerogen; water–oil interaction; Re-Os geochronology
INTRODUCTION
Petroleum systems govern key processes that promote the formation of oil and gas fields in sedimentary basins (Fig. 1; Magoon and Dow 1994). The preference of Re and Os for organic matter results in elevated abundances in organic-rich shales and carbonates compared with other crustal rocks (see Toolkit Fig. 5). This makes the Re-Os system suitable for tracing oil formation mechanisms and migration paths of hydrocarbons in time and space. In sedimentary basins, prolonged heating upon burial and subsidence induces a series of chemical transformations that alter the organic remains preserved in the sediments into substances of immediate practical use for our society— crude oil and natural gas. The initial requirement is the presence of a source rock rich in organic matter of the type that can produce oil or gas. Hydrocarbon generation begins with slow transformation of the organic remains into kerogen—a solid mixture of complex large heteroatomic organic molecules that are insoluble in organic solvents. As the rock is heated further, some of the kerogen is turned into very viscous bitumen, liquid oil, natural gas, or solid hydrocarbons (pyrobitumen) through a process called “thermal cracking.” Thermal cracking involves breaking bonds in the solid kerogen to form simpler molecules like alkanes (linear and branched saturated hydrocarbon chains or rings) and aromatics (hydrocarbons with a ring structure containing double or triple carbon bonds). The liquid and gaseous products accumulate in the pores of the source rock and start to freely flow only when individual molecules connect into droplets and subsequently form networks that allow oil to be expelled into adjacent rocks (primary migration). The expelled hydrocarbons move through permeable layers (secondary migration; Fig. 1) driven by buoyancy, the push from continuing hydrocarbon production and regional groundwater flow, and structural features in the host rock that create pressure differentials. Hydrocarbon seeps are the surface expression of this process, whereas subsurface oil reserves only accumulate when the migration path is blocked by an impermeable layer (seal) or steered and trapped by geologic structures such as faults (Fig. 1). The present-day composition of the oil is affected by chemical, physical, and given the right conditions, biological processes (biodegradation), including precipitation of heavy oil components, such as pyrobitumen and tar mats, and water washing of light hydrocarbons along migration paths.
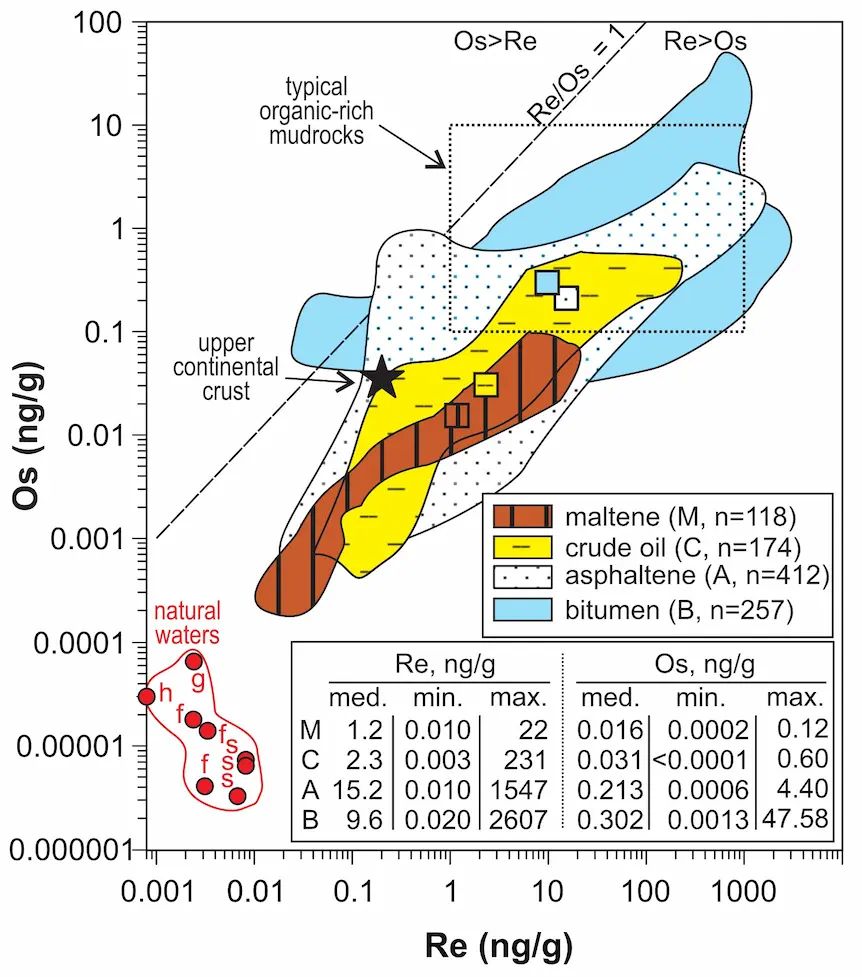
The Re-Os isochron method provides a unique view into the complex geologic history of petroleum systems (see Toolkit Figs. 1, 5, and 6). Pyrolysis experiments that mimic natural oil generation show that small portions of Re and Os are transferred from kerogen to the crude oil during thermal cracking (Rooney et al. 2012; Cumming et al. 2014). The effect on the metal budget of the rock is relatively minor, as typical source rocks have 2–3 orders of magnitude higher concentrations (100s ng/g Re, up to 10s ng/g Os) than crude oils with median contents of 2.3 ng/g Re and 0.03 ng/g Os (Fig. 2). Most of the Re-Os budget in source rocks is contained in kerogen, making up 5%–20% of the source rock, indicating concentrations in the 1000s ng/g Re and 100s ng/g Os. Metals in oil are therefore inherited from their source kerogens, which opens the possibility for Re-Os dating of hydrocarbon formation and for oil–source rock and oil–oil isotopic fingerprinting (e.g., Selby and Creaser 2005; Georgiev et al. 2016, 2019).
Throughout the geologic lifecycle of petroleum systems, most organic components and their Re and Os cargo are variably accompanied by waters, another important fluid in sedimentary basins. These interactions start early on as dissolved Re and Os species in seawater are reduced and adsorbed onto organic matter and buried into the sediment (Kendall et al. 2025 this issue). Water may also facilitate thermal cracking and helps drive the expulsion and migration of oil into the reservoir (Lewan 1994), where hydrocarbons accumulate on top of formation water and the two fluids remain in contact with each other for long periods of time (Fig. 1).
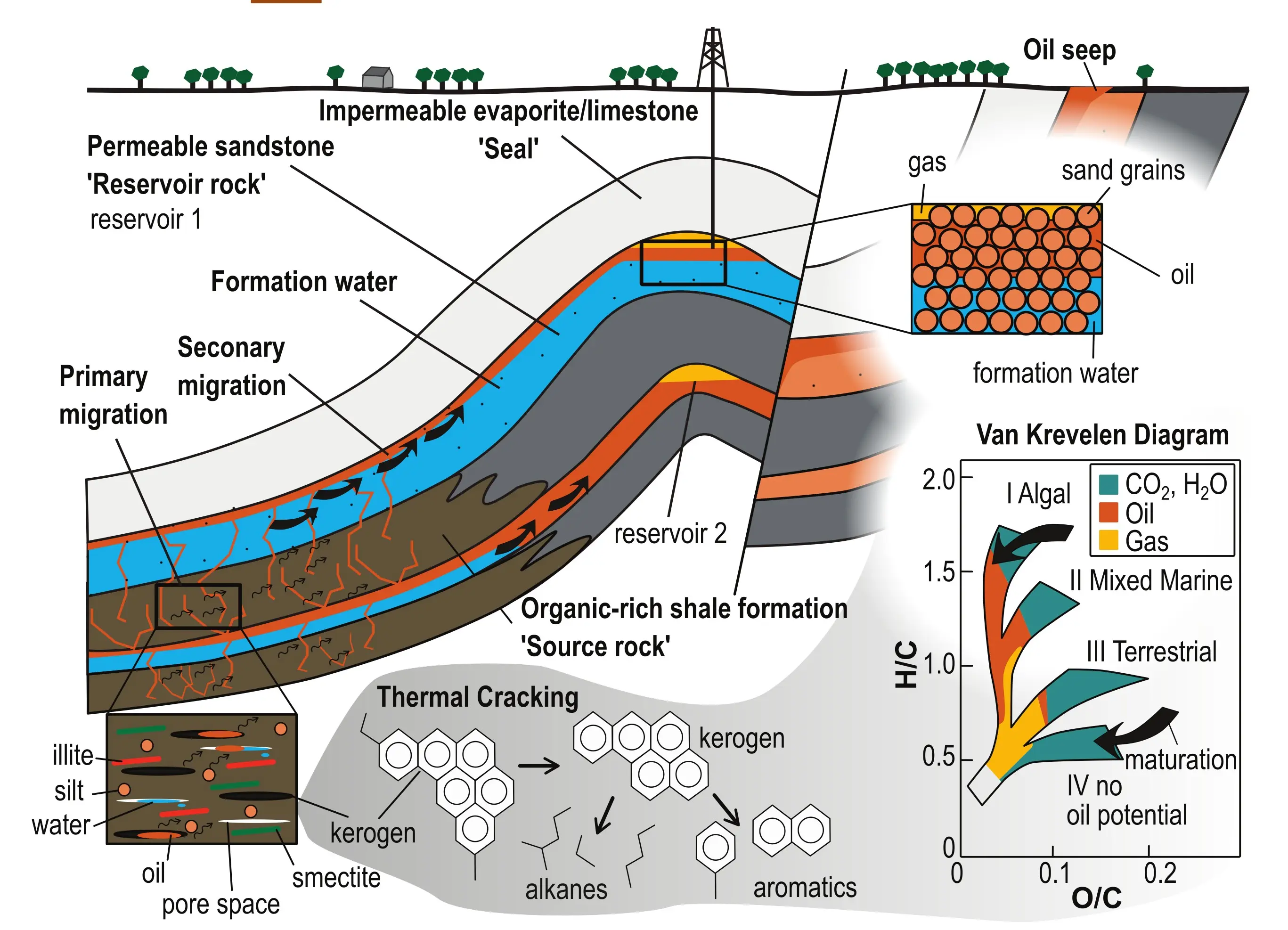
Download high-resolution image
Waters have 3–4 orders of magnitude lower Re and Os contents than oils (Fig. 2), which indicates that isotopic overprinting is only achieved through prolonged interaction that effectively transfers Re and Os into the oils. As water and oil usually have contrasting Os isotopic ratios, these interactions can leave a traceable mark on the isotopic composition of affected oils (e.g., Finlay et al. 2010; Hurtig et al. 2019; Georgiev et al. 2021).
Here we summarize the Re-Os contents and isotopic compositions of petroleum and its fractions, and those of natural waters. We further highlight fundamental knowledge gained from water–oil interaction experiments and the implications for interpreting Re-Os signatures in natural systems. Finally, we provide examples for application of Re-Os geochronology and isotope tracing to petroleum systems and a brief outlook on future new directions.
RE-OS IN PETROLEUM SAMPLES
The Re and Os contents (0.03–3000 ppb Re and 0.0004–50 ppb Os) in bitumen and crude oils are correlated, often elevated relative to typical crustal values (see Toolkit Fig. 5), and increase from light, low-density oils to heavier, more viscous oils to bitumen (Fig. 2). Crude oil is a complex mixture of many different hydrocarbons, which for practical purposes, are subdivided into two solubility classes: asphaltene (insoluble in a given alkane solvent) and maltene (soluble in the same alkane solvent). Asphaltenes comprise large, polar, aromatic, and structurally complex, heteroatomic molecules rich in C, S, N, and O and poor in H. In contrast, maltenes contain simpler hydrocarbons that are less polar and less aromatic with higher H/C ratios. Because asphaltene and maltene are not defined molecules but solubility classes, their physical and chemical properties systematically vary with the solvent used for asphaltene precipitation (Georgiev et al. 2016; DiMarzio et al. 2018). For example, the amount of asphaltene precipitated from an oil decreases with the use of progressively heavier solvents, such as the series of pentane (C₅H₁₂), heptane (C7H16), and decane (C10H22) (Fig. 3), whereas their Re and Os contents systematically increase (DiMarzio et al. 2018; Hurtig et al. 2020).
Rhenium and Os are hosted dominantly in the asphaltene fraction of crude oils (Fig. 2; Selby et al. 2007; Georgiev et al. 2016; Liu et al. 2019) and specifically in the most polar and aromatic asphaltene sub-fractions, where they follow most closely the distribution of Mo and Cd, and to a lesser extent that of V and Ni (DiMarzio et al. 2018). These observations suggest that the two metals are likely stored within large organic molecules with a functional group that binds to a central metal atom, such as porphyrins and non-porphyrin heteroatomic compounds. Within the maltene, Re and Os are similarly enriched in the most polar molecules, with aromaticity playing a secondary control (DiMarzio et al. 2018).
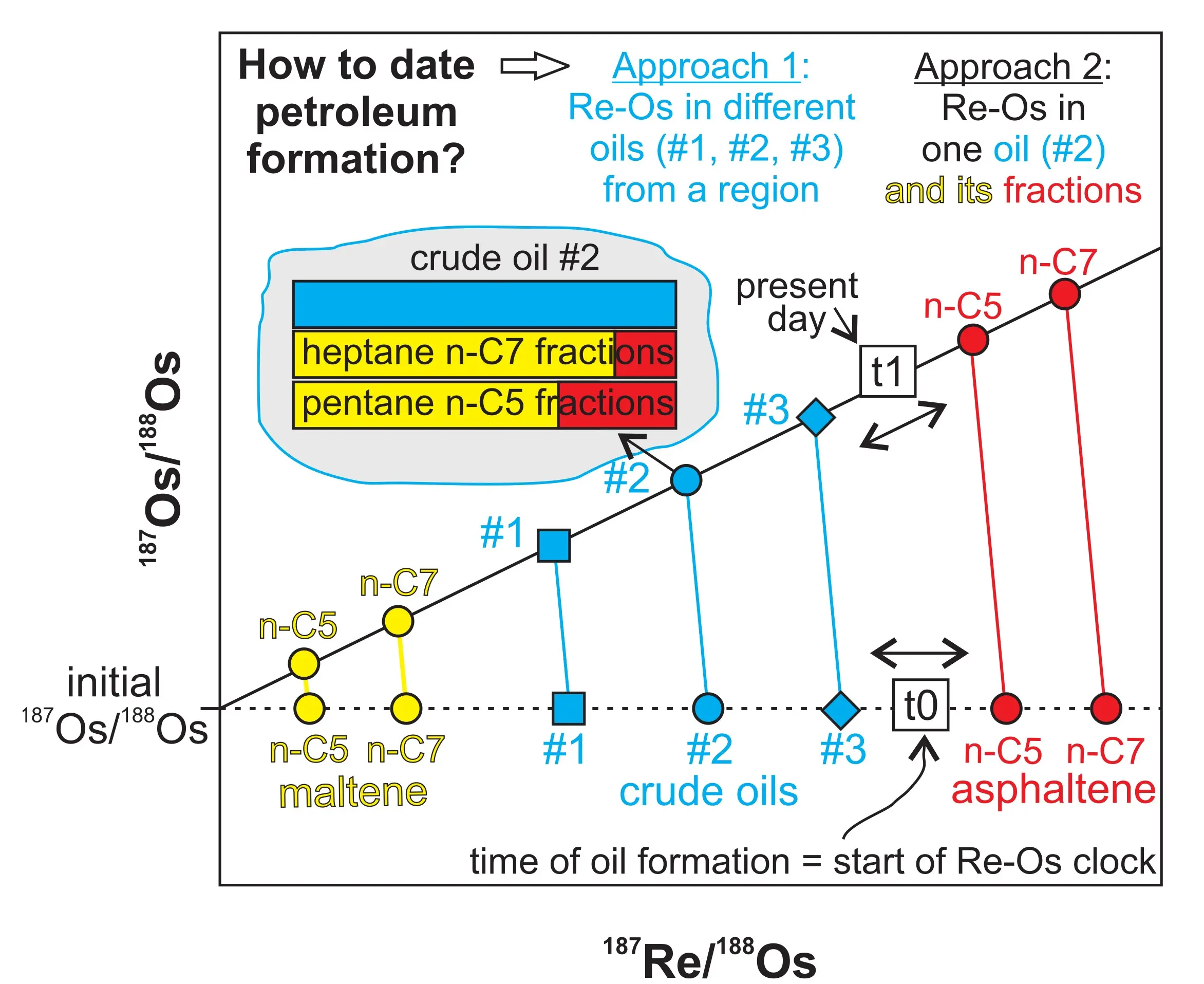
Accurate and precise Re-Os isotopic measurements of petroleum samples are facilitated by the elevated Re and Os contents in certain hydrocarbons (Fig. 2) and improvements in mass spectrometry, sample digestion, and chemical procedures (see Toolkit Fig. 3; Georgiev et al. 2016; Yang et al. 2020). Apparent Re-Os ages can be obtained by i) using multiple crude oil and/or asphaltene or bitumen samples from the same oil field, or ii) using a single crude oil sample and its asphaltene and maltene fractions (Fig. 3). In the first approach, the isotopic composition of several samples from a petroleum field or, more typically, a petroleum region are plotted on an isochron diagram to determine the age of the samples and their initial Os isotopic composition.
Download high-resolution image
Download high-resolution image
The first such application dated by Re-Os metal-rich heavy oils leached from West Canadian tar sands to 112 ± 5.3 Ma (Selby and Creaser 2005). Subsequent studies targeted the metal-rich asphaltene fraction of crude oils from specific geological regions. For example, Finlay et al. (2010) reported a Re-Os age of 147 ± 13 Ma for UK North Sea oils, and Lillis and Selby (2013) found a Triassic Re-Os age of 211 ± 13 Ma for oils from the Bighorn Basin, USA.
The second approach, analyzing different fractions derived from a single crude oil, is based on the recognition that asphaltene, crude oil, and maltene have different, and systematically varying, isotopic compositions. Typically, 187Re/188Os and 187Os/188Os ratios increase from maltene to crude oil to asphaltene (Fig. 3); regression of these data points on an isochron diagram yields the age of the oil and its initial 187Os/188Os. This approach was developed and used for the first time by Georgiev et al. (2016) to date heavy oils from Sicily at 27.5 ± 4.6 Ma, which coincides with the onset of regional collision that facilitated production and migration of hydrocarbons. The spread of asphalteneoil- maltene triplets on the isochron diagram can further be improved by using different solvents for asphaltene precipitation (Fig. 3). Subsequent studies have confirmed the systematic isotopic offset between oil fractions and the possibility of extracting an apparent Re-Os age from asphaltene-oil-maltene triplets (Georgiev et al. 2019; Liu and Selby 2019; Hurtig et al. 2020).
In both approaches, the degree of Os isotopic homogeneity of the oils and their fractions at the time zero is crucial for Re-Os geochronology; samples with initially heterogeneous isotopic compositions will adversely affect the precision and sometimes the accuracy of Re-Os isochron ages. These Re-Os formation ages for petroleum samples can be cross-checked with estimates for the timing of oil generation from basin burial models. Oil–rock and oil– water interactions, among other secondary processes, can modify the isotopic composition of oils and complicate the interpretation of Re-Os ages. Such challenges, however, present opportunities for identifying and dating secondary modification events. For example, altered oils from the Bighorn Basin yield a Miocene age, suggesting resetting by in-reservoir thermochemical sulfate reduction (Lillis and Selby 2013). Such resetting is not universal, as similarly altered bitumen from the Sichuan Basin in China yields a Re-Os age consistent with oil generation (Chu et al. 2023).
RE-OS IN WATER SAMPLES
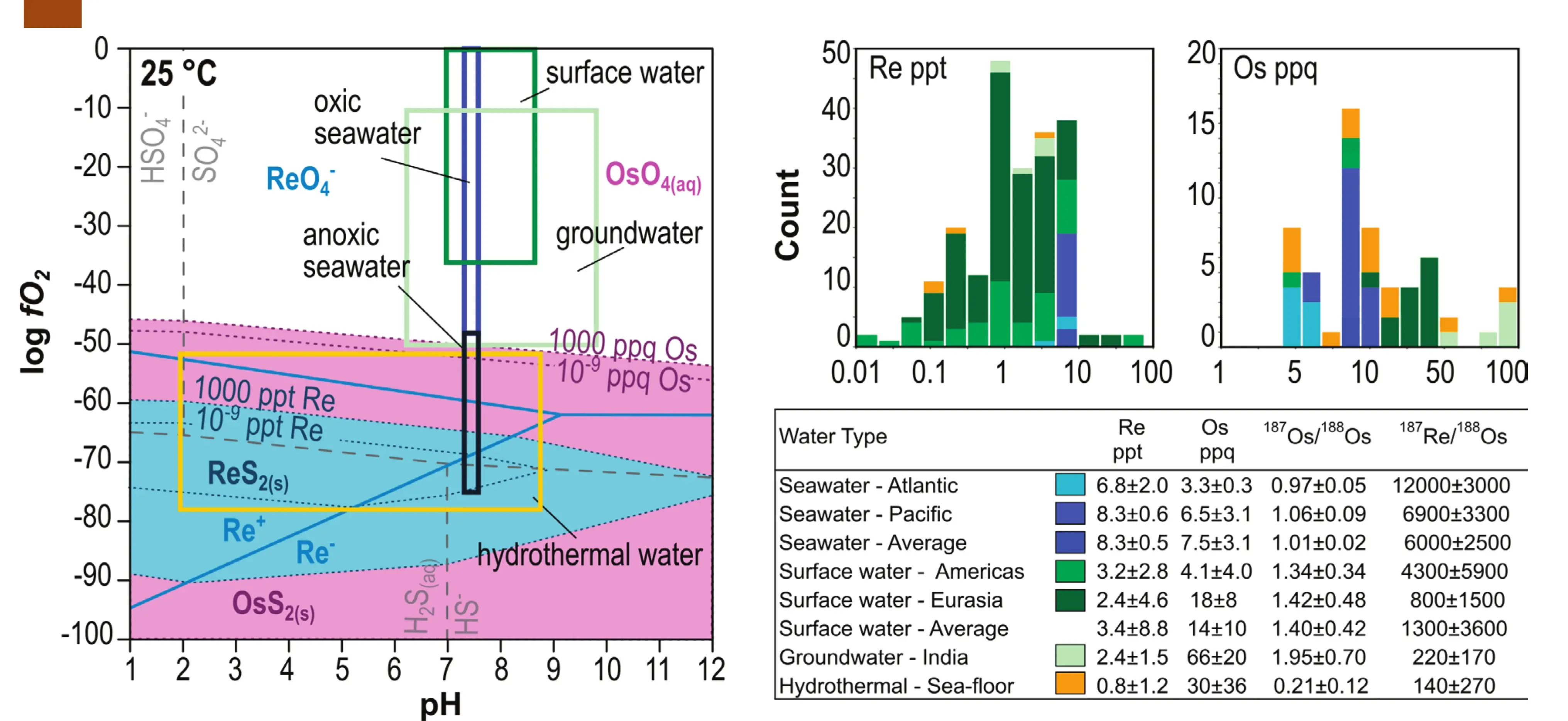
In aqueous solutions, Re and Os form hydroxyl, thiosulfate, and chloride complexes with a range of oxidation states (Yamashita et al. 2007; Helz 2022; Ghazi et al. 2024). At oxidizing conditions, the rhenate Re(VII)O4 − species dominates; at more reducing conditions, thioperrhenate Re(VII)OnS4-n − species become important; and at highly reducing conditions, the aqua ions Re(I)+ and Re(−I)− are stable (Fig. 4). Osmate Os(VIII)O4(aq) is the main Os species and little is known about Os chloride, thiosulfate, and aqua ion species. The solubilities of Re and Os in water are controlled by the stability of Re and Os sulfides and oxides, which are exceedingly rare close to the Earth’s surface. Thus, these two metals are usually incorporated as trace amounts in sulfides (e.g., pyrite, pyrrhotite, molybdenite), oxides (birnessite, vernadite, goethite), or organic matter (see Toolkit Fig. 4), where they occur in tetra- and trivalent oxidation states (Yamashita et al. 2007). Under oxidizing conditions, Re and Os are mobile as hydroxyl complexes (Fig. 4) and show median concentrations of 1.4 ppt Re (0.001 ng/g) and 9.4 ppq Os (9 × 10−6 ng/g) in natural water, which are 4–6 orders of magnitude lower than their median concentrations in crude oils (Fig. 2). Osmium solubility in water is more redox sensitive than that of Re (Fig. 4). Furthermore, Os, unlike Re, can incorporate into oxide and hydroxide minerals even under relatively oxidizing conditions, leading to low 187Re/188Os ratios (~1) in sea-floor nodules and more effective pathways for Os removal during sedimentation (Yamashita et al. 2007).
Surface waters (rivers, lakes, and estuaries) show average concentrations of 3.3 ppt Re and 14 ppq Os (Colodner et al. 1993; Sharma et al. 1999; Ghazi et al. 2024). The highest Re contents, measured in Andean rivers in Venezuela and lakes in Canada, are associated with Re leached from mine waste (Ghazi et al. 2024). The highest Os concentrations, attributed to weathering of shales, are measured in Himalayan rivers (Sharma et al. 1997) and show elevated 187Os/188Os ratios of 1.40 ± 0.42. Groundwaters have lower average Re contents of 3.4 ppt and comparatively higher average Os concentrations of 66 ppq with highly radiogenic 187Os/188Os ratios of 1.95 ± 0.70 (Colodner et al. 1999; Paul et al. 2010). In surface waters and groundwaters, elevated Os contents are correlated with radiogenic Os isotope signatures due to increased weathering of shales and fluid–rock reaction with organic-rich sedimentary rocks with high 187Os/188Os ratios (>1).
Seawater has average concentrations of 8.3 ppt Re and 7.5 ppq Os, with an average 187Os/188Os ratio of 1.00 ± 0.02 (Fig. 4). Rhenium concentrations show little to no variation in the seawater column with depth (Colodner et al. 1993; Ghazi et al. 2024), whereas Os concentrations vary considerably due to Os removal during fresh water–seawater mixing and reduction due to biological activity (Sharma et al. 1997). Preferential removal of Os in the seawater column can lead to variable Os concentrations of 2.8–10 ppt and significant variations in the 187Re/188Os
ratios, averaging at 6000 ± 2500.
Download high-resolution image
However, the Os isotopic composition of seawater is not impacted by these processes and changes only due to varying input from riverine, cosmogenic, and volcanic sources, thus providing a link to large-scale tectonic processes and climate change with time (see Toolkit Fig. 12; Kendall et al. 2025 this issue). Sea-floor hydrothermal fluids (~300 °C) show average concentrations of 0.8 ppt Re and 30 ppq Os with low 187Os/188Osratios of 0.21 ± 0.12 (Sharma et al. 1997; Miller et al. 2010). Elevated Os concentrations in sea-floor hydrothermal fluids are correlated with low 187Os/188Os ratios, indicating a mantle source for the fluids (Luguet et al. 2025 this issue) or extensive interaction with basalt and gabbro. Rhenium is not mobile in the plume and is rather incorporated into the sea-floor sulfides due to the reducing conditions and interaction of the fluid with mafic rocks. Sea-floor hydrothermal fluids, therefore, have low 187Re/188Os ratios of 130 ± 270.
RE-OS RESETTING DURING WATER-OIL INTERACTION
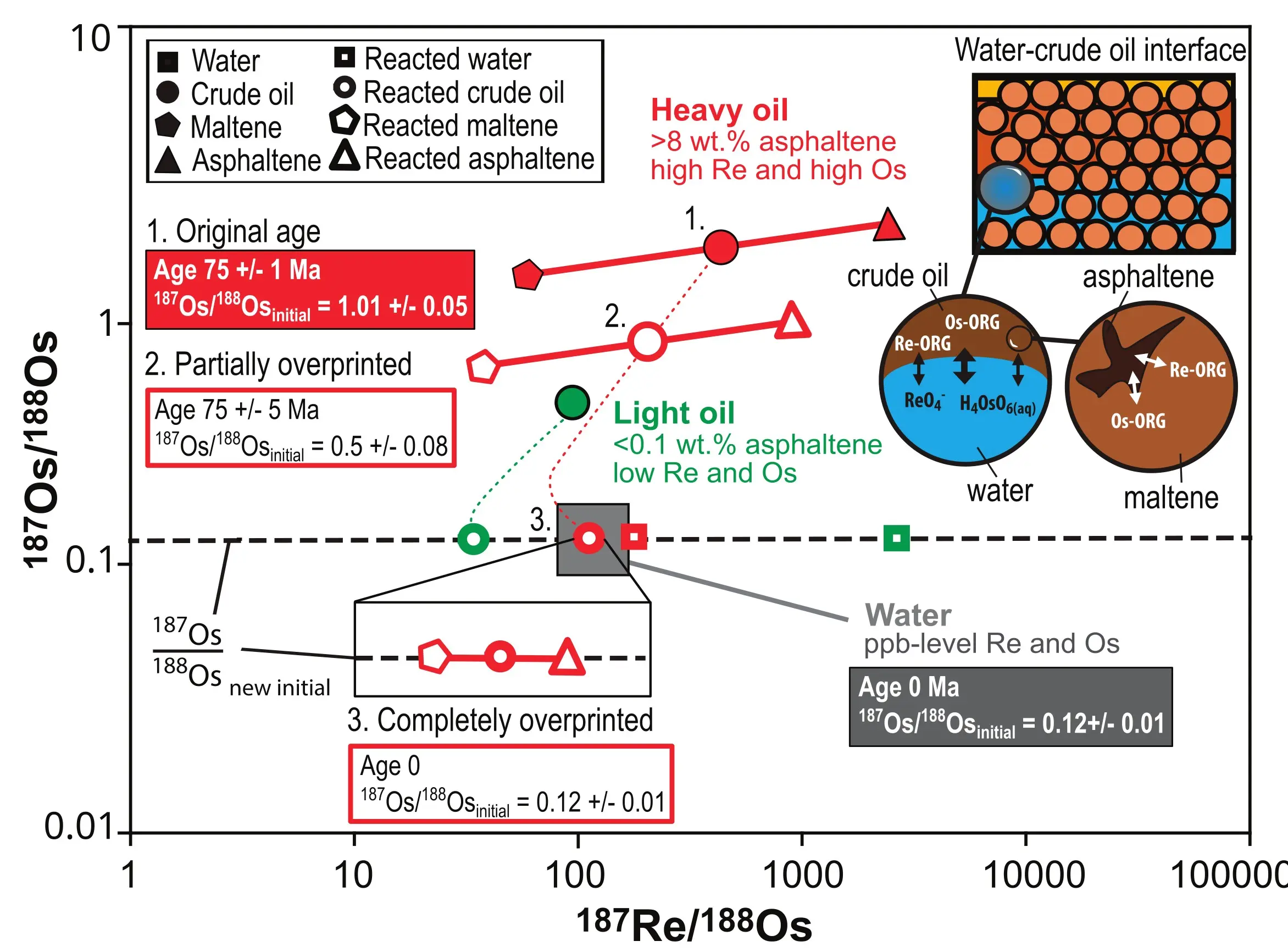
Water–oil interactions in petroleum systems (Fig. 1; Finlay et al. 2010; Georgiev et al. 2021) provide ample opportunities for Re and Os exchange between these two phases, which may reset the Re-Os clock in petroleum as demonstrated experimentally (Mahdaoui et al. 2015; Hurtig et al. 2019). At high Re and Os concentrations (ppm) in the experimental solutions, Re and Os strongly partition into the oils due to their organophile nature and reach saturation concentrations at ~400 μg/g (ppm) in oils with 20 wt.% asphaltene (Mahdaoui et al. 2015). At lower Re and Os concentrations (ppb to ppt), Os strongly partitions into the oils, whereas Re remains mobile as rhenate in the water (Hurtig et al. 2019). If the 187Os/188Os ratio is only partially overprinted through water–oil interaction, at all but the greatest extents of overprinting, the slope of the original asphaltene-oil-maltene triplet, i.e., the Re-Os age, is preserved (Fig. 5; Hurtig et al. 2019). In completely overprinted oils, the 187Os/188Os ratio of the reacted oil is the same as that of the water (Fig. 5), whereas the 187Re/188Os ratios are variable and depend on the type of crude oil. Heavy viscous oils with high asphaltene contents show a greater affinity for Re and Os and therefore obtain a similar or higher 187Re/188Os ratio compared with the water. Light, low-density oils with low asphaltene contents have a low affinity for Re and therefore obtain lower 187Re ratios compared with the original water composition (Fig. 5). Because of the complete overprinting of the Os isotopic composition, the age is effectively reset to zero. Collectively, these experiments underpin our understanding of how Re and Os are redistributed in organic materials that are in prolonged contact with water and the impact of this process on the Re-Os clock in oils.
Several case studies document these intimate conversations between oil and water. Low 187Re and 187Os ratios in crude oils from the Miller, Ninian, and Thistle oil fields, UK Atlantic Margin (Finlay et al. 2010) and the Solveig oil field, Norwegian North Sea (Georgiev et al. 2021) indicate interaction with natural waters. These oils are expected to have high 187Re/188Os and radiogenic 187Os/188Os
ratios inherited from their Jurassic–Cretaceous source rocks. The low 187Os/188Os ratios of 0.17–0.48 of these crude oils and their fractions are therefore inconsistent with the timing of oil formation and with the Re-Os isotopic composition of the source rock. Instead, low 187Os/188Os are explained by interaction with hydrothermal fluids carrying low 187Os/188Os (Fig. 4). These fluids likely have Re-Os isotopic compositions similar to those of sea-floor hydrothermal fluids characterized by low 187Os/188Os and low 187Re/188Os (Fig. 4). The resetting mechanism through water–oil interaction and its resulting Os isotopic compositions can be modeled using binary mixing revealing locally unaffected oil portions that still carry the Re-Os isotopic fingerprint tied to the formation age of the oil and its source rock.
PETROLEUM SYSTEMS: RE-OS APPLICATIONS
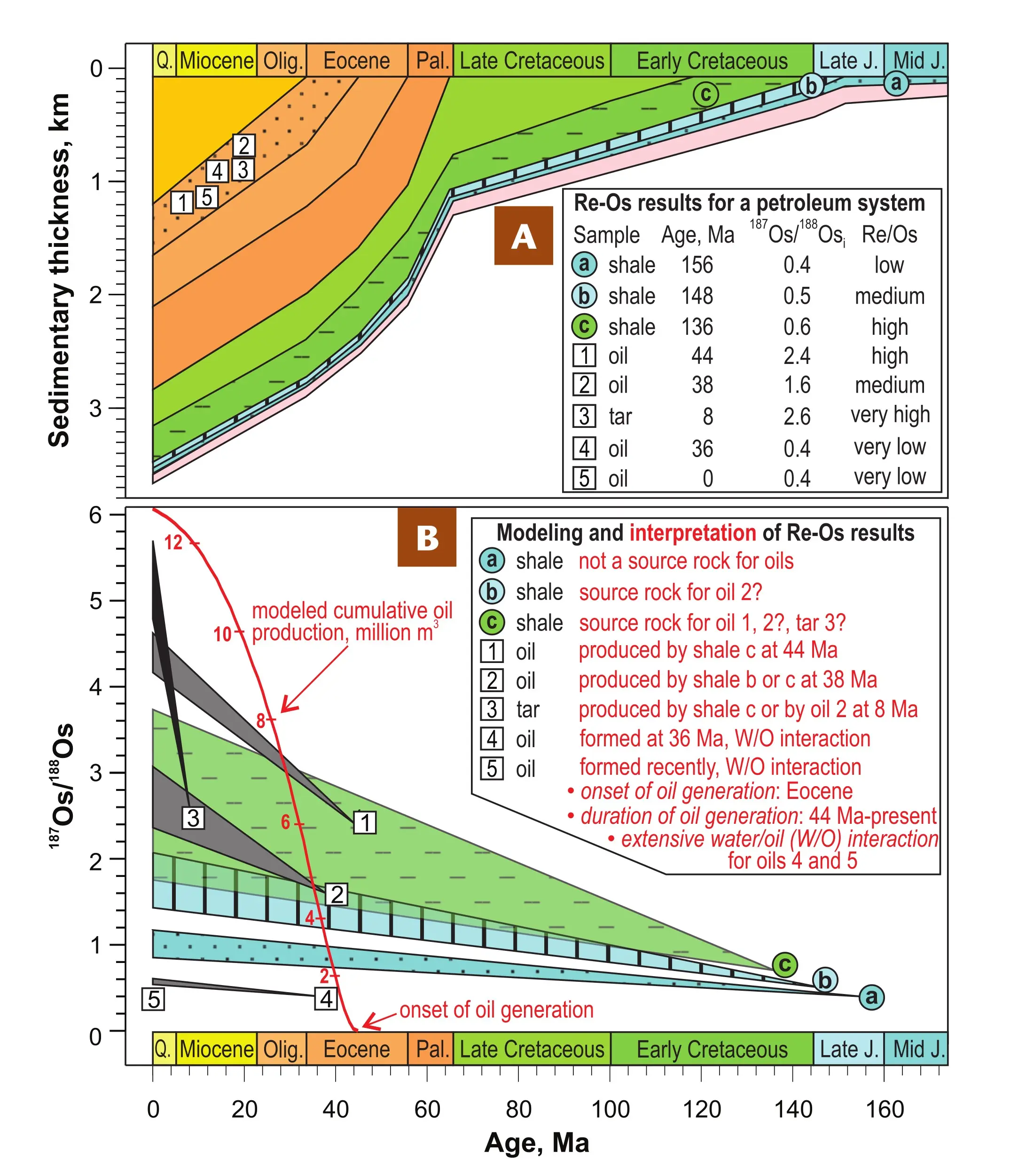
The Re-Os isotopic system provides valuable information on the genetic and temporal links between the different system components and the processes that shaped the present-day petroleum system (e.g., Selby and Creaser 2005; Finlay et al. 2011; Georgiev et al. 2019). These correlations are established by modeling the isotopic ratios of each sample back in time, based on the known age and the initial Os isotopic composition derived from the isochron regression of the samples in question (Fig. 6). To reconstruct genetic and temporal evolution of a petroleum system, the isotopic composition of its multiple components at present day and back in geologic time needs to be defined and put into context.
Download high-resolution image
This concept is illustrated based on case studies in the North Sea (Fig. 6; Georgiev et al., 2021). The North Sea petroleum system comprises three possible source rocks of loosely defined Middle Jurassic, Late Jurassic, and Early Cretaceous age, and four different oils and one tar mat (solid bitumen) of unknown origin and age encountered in Oligocene sandstone reservoirs. The burial history of the region suggests continuous oil generation from the mid-Eocene to present-day (Fig. 6). By dating all components (Fig. 6A inset bottom right) and modeling their 187Os/188Os ratios back in time to when they formed (Fig. 6B), we can test the possible genetic links between the potential source rocks, the oils, and the tar mat. The absence of an overlap of the modeled 187Os/188Os of shale (a) indicates that it cannot be the source for oils 1–3. Conversely, shales (b) and (c) show overlapping modeled 187Os/188Os with oil 2 at 38 Ma, the time of oil 2 generation. Shale (c) is the likely source of oil 1, as it is the only rock that had high enough 187Os/188Os at 44 Ma, the time of oil 1 generation. The 8 Ma bitumen (tar mat3) could have been produced either by shale (c), or by asphaltene precipitation from oil 2. The remaining oils 4 and 5 with Re-Os ages of 36 Ma and 0 Ma, respectively, have very low present-day and initial 187Os/188Os ratios that are indicative of isotopic modification of the oil, likely by substantial water–oil interaction. In the case of oil 4, the preservation of an isochron slope results from partial isotopic homogenization (Fig. 5) and the age likely represents the age of oil generation. For oil 5, the lack of a slope (age = 0) indicates either recent and ongoing oil generation with geologically rapid Os-isotopic overprinting by water–oil interaction, or complete isotopic overprint of the oil age by recent water–oil interaction. Other applications of the Re-Os clock to petroleum systems not illustrated on Figure 6 include dating the age of thermochemical sulfate reduction within an oil reservoir (Lillis and Selby 2013), dating reservoir rocks containing indigenous organic matter by first removing the oil phase (Georgiev et al. 2019), and identifying isotopically, and hence genetically, different hydrocarbon components (Georgiev et al. 2019). In addition, the Re-Os method is used for dating gas fields through Re-Os geochronology of solid bitumen remaining after a former oil charge was thermally cracked to produce the gas (e.g., Chu et al. 2023) and for identifying recent in-reservoir mixing of oil charges based on reverse isotopic relations in the oil fractions (Georgiev et al. 2021).
OUTLOOK
The Re-Os method has diverse applications to petroleum systems. As highlighted here, these include not only determining the age of different components of the system but also reconstructing their interaction in time. Important advances in analytical instrumentation (e.g., use of amplifiers equipped with 1012–1013 Ω resistors) and Re and Os extraction techniques (Yang et al. 2020) have overcome technical challenges and enabled new applications on a wider range of natural materials, including light oils, maltene components, and waters. The emerging use of matrix-matched reference materials for petroleum samples (Hurtig et al. 2020) and interlaboratory comparisons (Georgiev et al. 2018) are key for further improvements in the precision and accuracy of the method and to diversify its applications to petroleum systems.
Download high-resolution image
Integrative approaches combining apparent Re-Os ages with basin burial models and biomarker data provide a more holistic understanding of petroleum systems. The Re-Os method is also applicable in studies of crude oils that have lost their biomarker information as a result of biodegradation or extensive water-washing. Alone, or preferably combined with other trace metal systematics, Re-Os data can be used for oil–source and oil–oil fingerprinting and potentially identifying water–oil interactions linked to hydrocarbon migration pulses in basins. Employing Re-Os systematics in hydrocarbons linked to sedimentary ore deposits, such as Pb-Zn Mississippi Valley type (MVT) and Pb-Zn-Ag clastic-dominated, sedimentary-hosted deposits (CD, formerly SEDEX), could provide crucial new information on ore-forming mechanisms, fluid migration paths, and timing of ore formation. A benchmark study by Selby et al. (2005) showed that the bitumen age of the Polaris MVT Zn-Pb deposit coincides with the Rb-Sr age in sphalerite and paleomagnetic dating of ore formation. Another more recent study on Carlin-type Au deposits in the Nanpanjiang Basin in China also shows that ore formation coincides with hydrocarbon migration (Ge et al. 2021). Hydrocarbons not only concentrate Re and Os by several orders of magnitude, but can also concentrate Pb, Zn, Cu, Ag, and other metals as a result of water–oil interaction and initiate ore mineral precipitation (Hurtig et al. 2018). Ore and petroleum geologists may at last be having longoverdue intimate conversations to utilize the full potential of geochemical fingerprinting in hydrocarbons.
ACKNOWLEDGMENTS
SVG and NCH acknowledge past generous support from the Norwegian petroleum industry through grants to H. Stein and J. Hannah, notably CHRONOS at University of Oslo, and SNSF grants 164980 and 174313 to NCH. SVG acknowledges ongoing support from the Bulgarian National Science Fund, grant КП-06-ДВ/6. We thank Holly Stein and Laurie Reisberg for a constructive review of this article, as well as three external reviewers, Swapan Sahoo, Raymond Michels, and Robert Creaser. Nikola Botoucharov is thanked for feedback on Figure 1.
REFERENCES
Chu Z and 6 coauthors (2023) Re-Os dating of gas accumulation in Upper Ediacaran to Lower Cambrian dolostone reservoirs, Central Sichuan Basin, China. Chemical Geology 620: 121342, doi: 10.1016/j. chemgeo.2023.121342
Colodner D and 5 coauthors (1993) The geochemical cycle of rhenium: a reconnaissance. Earth and Planetary Science Letters 117: 205-221, doi: 10.1016/0012-821x(93)90127-u
Cumming VM, Selby D, Lillis PG, Lewan MD (2014) Re–Os geochronology and Os isotope fingerprinting of petroleum sourced from a Type I lacustrine kerogen: insights from the natural Green River petroleum system in the Uinta Basin and hydrous pyrolysis experiments. Geochimica et Cosmochimica Acta 138: 32-56, doi: 10.1016/j.gca.2014.04.016
DiMarzio JM, Georgiev SV, Stein HJ, Hannah JL (2018) Residency of rhenium and osmium in a heavy crude oil. Geochimica et Cosmochimica Acta 220: 180-200, doi: 10.1016/j.gca.2017.09.038
Finlay AJ, Selby D, Osborne MJ, Finucane D (2010) Fault-charged mantle-fluid contamination of United Kingdom North Sea oils: insights from Re-Os isotopes. Geology 38: 979-982, doi: 10.1130/g31201.1
Finlay AJ, Selby D, Osborne MJ (2011) Re-Os geochronology and fingerprinting of United Kingdom Atlantic margin oil: temporal implications for regional petroleum systems. Geology 39: 475-478, doi: 10.1130/g31781.1
Ge X and 5 coauthors (2021) Genetic relationship between hydrocarbon system evolution and Carlin-type gold mineralization: insights from Re-Os pyrobitumen and pyrite geochronology in the Nanpanjiang Basin, South China. Chemical Geology 559: 119953, doi: 10.1016/j.chemgeo.2020.119953
Georgiev SV and 6 coauthors (2016) Re–Os dating of maltenes and asphaltenes within single samples of crude oil. Geochimica et Cosmochimica Acta 179: 53-75, doi: 10.1016/j.gca.2016.01.016
Georgiev SV and 6 coauthors (2018) Comparison of chemical procedures for Re-isotopic measurements by N-TIMS. Chemical Geology 483: 151-161, doi: 10.1016/j.chemgeo.2018.03.006
Georgiev SV and 7 coauthors (2019) Comprehensive evolution of a petroleum system in absolute time: the example of Brynhild, Norwegian North Sea. Chemical Geology 522: 260-282, doi: 10.1016/j. chemgeo.2019.05.025
Georgiev SV, Stein HJ, Hannah JL, di Primio R (2021) Timing and origin of multiple petroleum charges in the Solveig oil field, Norwegian North Sea: a rhenium-osmium isotopic study. AAPG Bulletin 105: 109-134, doi: 10.1306/02272019219
Ghazi L and 5 coauthors (2024) The global biogeochemical cycle of rhenium. Global Biogeochemical Cycles 38: e2024GB008254, doi: 10.1029/2024gb008254
Helz GR (2022) The Re/Mo redox proxy reconsidered. Geochimica et Cosmochimica Acta 317: 507-522, doi: 10.1016/j.gca.2021.10.029
Hurtig NC, Hanley JJ, Gysi AP (2018) The role of hydrocarbons in ore formation at the Pillara Mississippi Valley-type Zn-Pb deposit, Canning Basin, Western Australia. Ore Geology Reviews 102: 875-893, doi: 10.1016/j.oregeorev.2018.09.012
Hurtig NC, Georgiev SV, Stein HJ, Hannah JL (2019) Re-Os systematics in petroleum during water-oil interaction: the effects of oil chemistry. Geochimica et Cosmochimica Acta 247: 142-161, doi: 10.1016/j.gca.2018.12.021
Hurtig NC and 6 coauthors (2020) Re-Os geochronology for the NIST RM 8505 crude oil: the importance of analytical protocol and uncertainty. Chemical Geology 539: 119381, doi: 10.1016/j. chemgeo.2019.119381
Kendall B, Creaser RA, Hannah JL, Goswami V, Tripathy G (2025) Reel-to-reel Re-Os records: Earth system transactions preserved in sediments. Elements 21: 264-270
Lewan MD (1994) Assessing natural oil expulsion from source rocks by laboratory pyrolysis. In: Magoon LB, Dow WG (eds) The Petroleum System – From Source to Trap. AAPG Memoir 60, Tulsa, pp 201-210, doi: 10.1306/m60585c11
Lillis PG, Selby D (2013) Evaluation of the rhenium–osmium geochronometer in the Phosphoria petroleum system, Bighorn Basin of Wyoming and Montana, USA. Geochimica et Cosmochimica Acta 118: 312-330, doi: 10.1016/j.gca.2013.04.021
Liu J, Selby D, Zhou H, Pujol M (2019) Further evaluation of the Re-Os systematics of crude oil: implications for Re-Os geochronology of petroleum systems. Chemical Geology 513: 1-22, doi: 10.1016/j. chemgeo.2019.03.004
Luguet A, Widom E, Liu J (2025) The osmium isotope perspective on the dynamics of the post-Archean mantle. Elements 21: 283-289
Magoon LB, Dow WG (1994) The petroleum system. In: Magoon LB, Dow WG (eds) The Petroleum System – From Source to Trap AAPG Memoir 60, Tulsa, pp 7759-7767, doi: 10.1306/m60585c1 Mahdaoui F, Michels R, Reisberg L, Pujol M, Poirier Y (2015) Behavior of Re and Os during contact between an aqueous solution and oil: consequences for the application of the Re–Os geochronometer to petroleum. Geochimica et Cosmochimica Acta 158: 1-21, doi: 10.1016/j.gca.2015.02.009
Miller CA, Peucker-Ehrenbrink B, Walker, BD, Marcantonio F (2011) Re-assessing the surface cycling of molybdenum and rhenium. Geochimica et Cosmochimica Acta 75: 7146-7179, doi: 10.1016/j. gca.2011.09.005
Paul M and 6 coauthors (2010) Dissolved osmium in Bengal plain groundwater: implications for the marine Os budget. Geochimica et Cosmochimica Acta 74: 3432-3448, doi: 10.1016/j.gca.2010.02.034
Rooney AD, Selby D, Lewan MD, Lillis PG, Houzay JP (2012) Evaluating Re–Os systematics in organic-rich sedimentary rocks in response to petroleum generation using hydrous pyrolysis experiments. Geochimica et Cosmochimica Acta 77: 275-291, doi: 10.1016/j.gca.2011.11.006
Selby D, Creaser RA (2005) Direct radiometric dating of hydrocarbon deposits using rhenium-osmium isotopes. Science 308: 1293-1295, doi: 10.1126/ science.1111081
Selby D, Creaser RA, Dewing K, Fowler M (2005) Evaluation of bitumen as a 187Re– 187Os geochronometer for hydrocarbon maturation and migration: a test case from the Polaris MVT deposit, Canada. Earth and Planetary Science Letters 235: 1-15, doi: 10.1016/j.epsl.2005.02.018
Selby D, Creaser RA, Fowler MG (2007) Re– Os elemental and isotopic systematics in crude oils. Geochimica et Cosmochimica Acta 71: 378-386, doi: 10.1016/j. gca.2006.09.005
Sharma M, Papanastassiou DA, Wasserburg GJ (1997) The concentration and isotopic composition of osmium in the oceans. Geochimica et Cosmochimica Acta 61: 3287-3299, doi: 10.1016/ s0016-7037(97)00210-x
Yamashita Y, Takahashi Y, Haba H, Enomoto S, Shimizu H (2007) Comparison of reductive accumulation of Re and Os in seawater–sediment systems. Geochimica et Cosmochimica Acta 71: 3458-3475, doi: 10.1016/j.gca.2007.05.003
Yang G and 6 coauthors (2020) Chemical procedures for rhenium extraction from geological samples: optimizing the anion resin bead clean‐up step. Geostandards and Geoanalytical Research 44: 231-242, doi: 10.1111/ggr.12318

