August 2025 Issue Table of Contents
1811-5209/25/0021-0256$2.50 DOI: 10.2138/gselements.21.4.256
Keywords: Re-Os geochronology; molybdenite; arsenopyrite; pyrite; El Teniente; Homestake, Hemlo
INTRODUCTION
In the beginning, there was molyb- denite (MoS2, molybdenum disul- fide), a mineral that few beyond ore deposit geologists would think about or even know. But molybdenite is in fact not so rare, and its importance as a critical host for rhenium (Re) has put it in the limelight. Molybdenite is the only naturally occur- ring mineral that carries significant Re (Fig. 1). Rhenium substitutes into the Mo cation site in molybdenite along a partial solid solution series with exceedingly rare rheniite (ReS2). Rhenium typically enters molybdenite at the ppm level and rarely reaches the percent level.
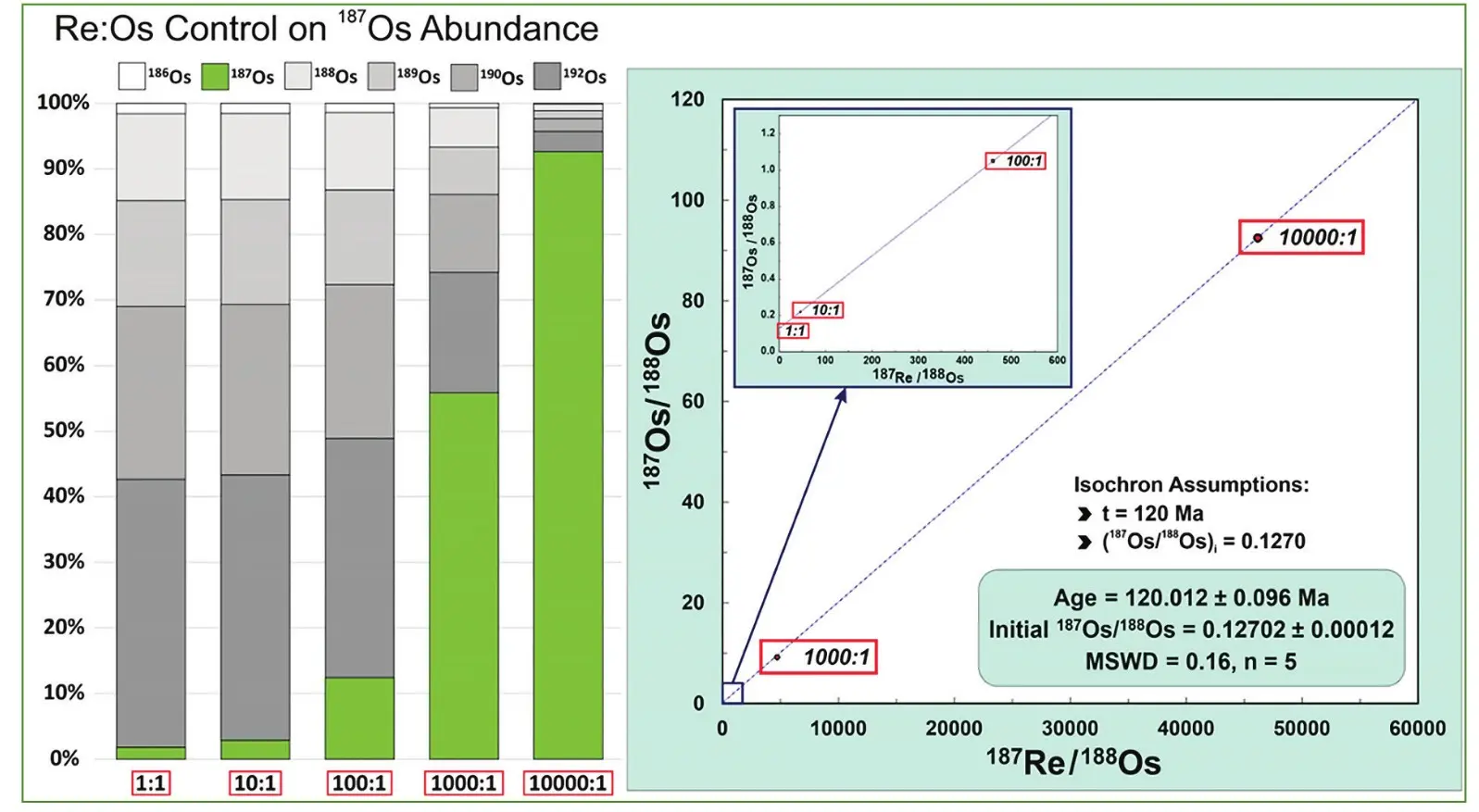
Molybdenite presents a very special case for the 187Re–187Os chronometer (see Stein references in Toolkit). In addition to significant Re, critically, molybdenite incorporates essentially no Os upon crystallization, such that all Os present is 187Os, the product of in situ decay of parent 187Re (Toolkit, Fig. 2). Thus, there is essentially no initial common Os to be consid- ered, greatly simplifying the age equation (compare equation with Toolkit, Fig. 6A). The molybdenite age (t) can be calculated from the following simple expression, where the 187Re decay constant is represented by λ: 187Os = 187Re (eλt − 1)
What could be so simple and yet so elegant? Molybdenite can be found in many felsic-silicic terranes, even where previously unreported, and in nearly all Cu-Au-Mo(-Ag) porphyry-style deposits (Fig. 3). Molybdenite is a common accessory mineral in Au deposits, in contact metamorphic aureoles, and in skarn- related ore deposits.

Download high-resolution image
Download high-resolution image

Download high-resolution image
It may be an accessory mineral in some granites, much like zircon or monazite, and can be found with other sulfides in shear zones. It forms in volcanic sublimates and geothermal systems. Molybdenite is widespread in some terranes, particularly Precambrian shields, even if in sparse quanti- ties, providing a tiny sentinel that records and retains metamorphic histories.
Molybdenite is assumed to be without measurable initial Os, although there were early attempts to explore the extremely small non-radiogenic Os content in molybdenite, among them the work of John Morgan (see Heritage, p. 291 this issue). The assumption of negligible common Os may be tested with implementation of an Os double spike (Markey et al. 2003).
A CHALLENGING OPENING ACT
The headwind in proving the integrity of the Re-Os chronometer in molybdenite was intense, as early results were highly criticized by some whose long-held ore deposit models began to unravel as new and uncomfortable Re-Os ages emerged. The advent of NTIMS combined with isotope dilution (see Toolkit, Fig. 3) allowed molybdenite to prevail, leaving little doubt that this chronometer was indeed remarkably robust (Raith and Stein 2000) and remained chemically unscathed during metamorphism and ductile deformation (Stein et al. 2002). The chronometer’s durability is largely tied to the chalcophile-siderophile nature of Re and Os, making molybdenite and other sulfides the overwhelming hosts for these elements in crustal silicate rocks. The extreme affinity of Re for molybdenite renders dating of other sulfides in the presence of molybdenite futile. In fact, when in contact with other sulfides, loss of a tiny fraction of radiogenically derived 187Os from the molybdenite does not perceptibly affect the molybdenite age but leads to wildly older ages for adjacent sulfides with ppb-level Re contents (Stein et al. 2003).
It quickly became evident that molybdenite Re-Os ages are primary and robust when compared with K-Ar or ⁴⁰Ar/³⁹Ar ages, which for decades were the favored methods for dating ore deposits. Unlike such ages, which are derived from gangue or alteration minerals (e.g., hydrothermal biotite, sericite, adularia), Re-Os ages are not a proxy but directly date mineralization (Suzuki et al. 1996; Watanabe and Stein 2000). The Re-Os clock is locked in upon molybdenite crystallization, and even osumilite-grade contact metamorphism (>900 °C) fails to disturb molybdenite Re-Os systematics (Bingen and Stein 2003). This discovery had far-reaching implications for determining not only the timing, but also the duration of events that created multiple superimposed ore bodies (e.g., porphyry systems). An undeserved fixation on how long it takes to create a porphyry-style ore deposit ensued, with estimates ranging from tens of thousands to millions of years. Ultimately, both Re-Os and argon-based chronologies have unique and complementary strengths, with Re-Os providing the absolute crystallization age and argon dating the cooling history of ore-bearing terranes, which arguably impacts duration of mineralization.
Historical Hats and Polytype Distraction
In the 1950s and 1960s, molybdenite, with its extraordinarily high Re concentrations and essentially pure 187Os content (negligible common Os), became the subject of study for a group of German and Swiss scientists, chief among them W. Herr and B. Hirt. Many of these early studies were carried out and published in association with the International Atomic Energy Agency (Vienna), leading to the first reliable estimates of the 187Re half-life at 43 ± 5 Gy. Using this half-life, the ages of about 30 molybdenites were determined with precisions ranging from about ±2% for an Archean sample from Greenland to about ±15% for a 38 Ma molybdenite from the USA. Molybdenites of presumably known age were then used to further refine the 187Re half-life (Hirt et al. 1963), but after 1967, no further molybdenite Re-Os age determinations were reported for 15 years.
In the 1970s, molybdenite studies turned toward measuring Re concentrations and polytype proportions in molybdenite, attempting to uncover a relationship between geologic setting and some identifiable molybdenite feature. A relationship between molybdenite’s two polytypes (hexagonal 2H1 and rhombohedral 3R) and geologic setting or deposit type was considered. Attempts to correlate Re concentration with temperature or polytype proved futile. While this polytype diversion garnered considerable interest, little progress was made in development of the Re-Os chronometer in molybdenite during the 1970s. Today, polytype remains an unsettled enigma, although new technology has provided enhanced insight into molybdenite’s complex structural heterogeneity.
The Alteration Altercation
To bring the Re-Os chronometer out of dormancy, Luck and Allègre (1982) redetermined the 187Re half-life as 45.6 ± 1.2 Ga, using the more precise secondary ion mass spectrometry (SIMS) method to date 11 molybdenites. Whereas some of their Re-Os ages agreed moderately well with ages based on other dating methods for surrounding rocks, others (e.g., Preissac, Canada; Climax, USA) were impossibly old (some greater than the Solar System!). Unfortunately, rather than considering possible complexi- ties in the chemical separation protocols for Re and Os, Luck and Allègre (1982) attributed their wildly aberrant ages to Re loss during metamorphism and/or hydrothermal alteration. The Re-Os chronometer in molybdenite, there- fore, was labeled unreliable.
A decade later at the University of Arizona (USA), workers used selected K-Ar ages to evaluate accuracy for new Re-Os ages from Arizona porphyry Cu deposits. On further study, these workers concluded that Re loss during hydrothermal alteration explained older Re-Os ages and recommended a series of imaging tests for molybdenites before dating (McCandless et al. 1993). Suzuki et al. (2000) experimen- tally studied the effects of hydrothermal alteration on molybdenite Re-Os systematics, and subsequently attrib- uted five incorrect Re-Os ages for the Galway granite to isotopic disturbance during hydrothermal fluid circulation (Suzuki et al. 2001). However, Selby et al. (2004) dated the same samples using ID-NTIMS and obtained accurate and reproducible ages. The issue appeared to be the analytical techniques (microwave digestion and ICP-MS) used by the Suzuki team. The use of imaging to evaluate a molybdenite for Re-Os dating was short-lived, as also was the notion that (sulfide-stable) hydrothermal alteration rendered Re-Os ages inaccurate. The tide of Re-Os ages just kept on rising. The ages made geologic sense, and they were reproducible.
FROM ALKALINE FUSION TO CARIUS TUBE DISSOLUTION
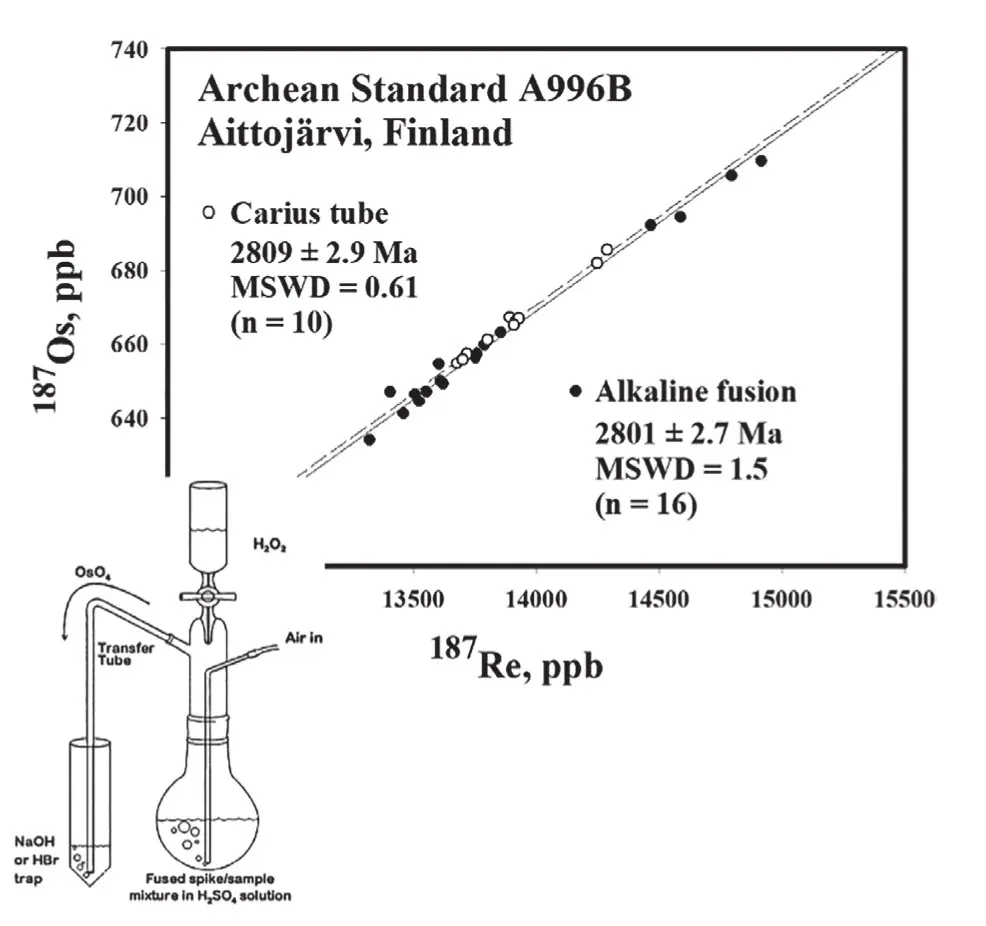
The alkaline fusion method was used for Re-Os dating in the early years, with the greatest concern being assurance of full sample-spike equilibration. The fusion procedure was long, tedious, and elaborate (Markey et al. 1998). Dissolution of the fusion product and ultimate isolation of Re and Os employed a delicate boiling flask apparatus used to transfer OsO4 into an HBr or NaOH trap (Fig. 4 inset). Introduction of the Carius tube method (see Reisberg and Stein 2025 this issue), in which sample-spike equilibration is achieved in a HNO3(-HCl) mixture in a sealed borosili- cate tube (held in a steel jacket at 240 °C for 12 h) was a welcome advance. The Carius tube method greatly reduced the potential for operator error, although there is some danger, as the tube pressure must be reduced using acetone–dry ice after heating and tubes must be opened with care.
With the changeover from alkaline fusion to Carius tube, it was critical to demonstrate that, despite the procedural complexities of the former, both methods produced like Re-Os ages (Fig. 4). Not surprisingly, the variability in alkaline fusion ages exceeds that of Carius tube ages.
Download high-resolution image
A new era opened for Re-Os analysis. The challenges endemically associated with open-system Re-Os alkaline fusion chemistry and operator variability slipped into the background, and sample-spike equilibration was assured with the introduction of the simpler and cleaner Carius tube technique. Streamlining the chemical procedure opened new possibilities, but several issues had to be addressed before the potential of the technique could be fully realized. These included reducing blanks (Yang et al. 2015), improving mass spectrometry measurements (Zimmerman et al. 2014), evaluating the accuracy of the 187Re decay constant, and standardizing the treatment of uncertainties among Re-Os laboratories.
Unspun Uncertainties
As Re-Os emerged as a powerful dating tool in the mid-1990s, AIRIE’s Re-Os ages for molybdenite from hightemperature skarn mineralization yielded ages consistently ~1.5% older than corresponding zircon U-Pb ages. Key to understanding this peculiarity was the high-temperature contact metamorphic setting and supporting petrography, suggesting zircon and molybdenite were contemporaneous. This puzzling and consistent mismatch was resolved with a significantly revised 187Re decay constant, still in use today (see Toolkit). A later study showed Re-Os ages for Cenozoic to Proterozoic molybdenites in agreement with zircon U-Pb ages, further affirming the Smoliar et al. 187Re decay constant value (Selby et al. 2007). The uncertainty on the improved 187Re decay constant, however, is about two times larger than for the U-Pb isotopic system. Nevertheless, when comparing Re-Os model ages among molybdenites, the uncertainty on the 187Re decay constant can be removed, as it is simply an error magnifier shared by all analyses. However, when comparing Re-Os ages with radiometric ages derived from other isotopic systems (e.g., U-Pb), it is essential that all ages include the decay constant uncertainty for their associated parent isotope.
Download high-resolution image
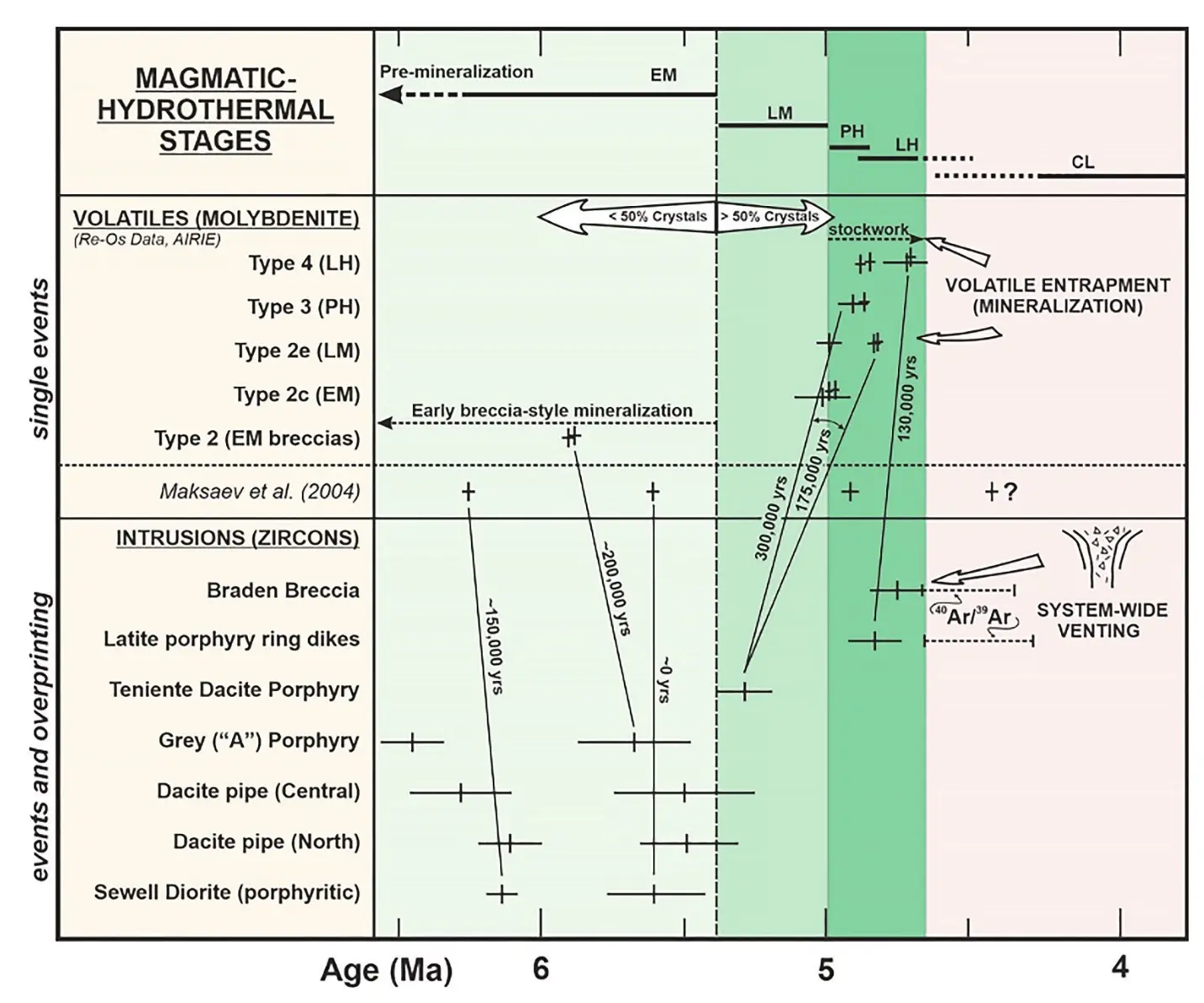
Employing the revised 187Re decay constant, the new mantra for evaluating Re-Os ages became a tight matchup with a presumed corresponding U-Pb zircon age. When the match was less than perfect, this gave an opening for the critics of Re-Os dating. We cannot assume, however, that U-Pb zircon ages should correspond to Re-Os ages under all, or even most, geologic circumstances. There is no better example than porphyry-style mineralization to confront this unfounded assumption (Fig. 5).
Multiple Re-Os ages from a single sample or site may reveal unexpected discrepancies that require thoughtful interpretation and evaluation in geologic context; none of the ages should be discarded for a “preferred age” based on agreement with a U-Pb age. The misalignment of U-Pb and Re-Os ages in porphyry systems is to be expected (Fig. 5). Multiple (explosive) pulses of volatile-rich fluid may travel far from their magmatic source to create molybdenitebearing stockwork veining that may be brittlely emplaced into older units (Fig. 3), trapped in magma mushes, and/ or expelled into the country rock. In contrast, magmatically crystallized zircon and inherited zircon cargos are common, even as hydrothermal zircon is also possible. In sum, molybdenite is generally the (late) product of volatilerich fluids at any scale, whereas zircon is most often associated with (early) magmatic crystallization. The U-Pb and Re-Os data in Figure 5 from the giant El Teniente porphyry Cu system (Chile) were the first to show non-matching ages for molybdenite-bearing veins and their zircon-bearing host rocks. Together with an early AIRIE study in 2002 at Los Pelambres (Chile), it was shown that several million years are needed to build a giant porphyry Cu deposit. Significantly, at Los Pelambres, it was shown that field designation of a vein type based on only morphology, mineralogy, and associated vein alteration could not be used to tell relative time of vein emplacement (see Stein reference in Toolkit). The economic geology community did not accept the new Re-Os ages for many years, as the ages demanded a re-examination of longheld models for porphyry-style mineral deposits.
It is not clear that laboratories publishing Re-Os data use the same rigor in calculating uncertainties, particularly errors associated with substantial over- or underspiking of samples, which can lead to enormous age uncertainty. Uncertainties that must be propagated into the precision of a Re-Os age include: (1) uncertainty of spike calibrations, (2) weighing errors (when using separate versus mixed Re and Os spike solutions), (3) uncertainty related to blank reproducibility (reported data should always be blank-corrected), and (4) uncertainty of mass spectrometer measurements. Blanket uncertainties should never be applied to all data, and an uncertainty should be calculated for each data point.
Certainty with Reference Materials
In the late 1990s, AIRIE adopted the HLP-5 milled powder from the Huanglongpu Mo-HREE deposit as an in-house reference material for tracking reproducibility. Multiple prepared HLP-5 separates were provided by Prof. Du Andao (cf. Heritage, p. 290 this issue) for inter-laboratory comparison. Many start-up Re-Os laboratories requested aliquots of this molybdenite, and it was easier to get them through AIRIE than through China. As a consequence, AIRIE’s supply began to run low, and another in-house reference material from a molybdenite-bearing leucosome at Aittojärvi (Finland) was adopted (Fig. 4). While sharing in-house reference materials was a first step, the community needed a certified NIST (National Institute of Science and Technology, USA) refer- ence material (RM) that could be globally accessed for true interlaboratory comparison. While at the United States Geological Survey (USGS), Holly Stein obtained a large sack of molybdenite powder from the Henderson Mill (Colorado, USA) which was the ore feed entering the mill on that day. Bringing the sack back to the USGS-Reston, she and John Morgan washed all reagents associated with the froth flota- tion process to recover a cleaned molybdenite. The NIST employed a robust procedure to physically mix and split the molybdenite, providing numbered bottles for testing by the only two laboratories that were successfully dating molybdenite in the early 2000s (Markey et al. 2007). The Henderson RM thus went to market as RM#8599 with an accompanying NIST certificate of investigation. The Henderson RM is for ages only, not for Re and Os concentrations, as paired concentrations of Re-Os vary slightly between analyses but remain coupled such that the 187Re–187Os age is reproducible.
Double Os Spike
The question of common Os in molybdenite came to the forefront when, while using individual 185Re and 190Os spike solutions, certain molybdenites were found to have significant common Os. Thus, the idea of using a single solution with 185Re–188Os–190Os spikes ensued (Markey et al. 2003). The mixed Re-double Os solution eliminates weighing errors, as deviation from true weight affects all spike isotopes equally. Not only could a more accurate determination of 187Os be made using the 188Osspike, but the 190Os spike could be used to accurately measure192Os, the most abundant Os isotope. Finally, the invariant (stable) 188Os / 190Os ratio provides an internal correction for instrumental mass fractionation. Thus, precise evaluation of the common Os in molybdenite could be routinely determined with the mixed Re-double Os spike. Laboratories not employing a mixed Re-double Os spike risk reporting Re-Os ages that are slightly, or even significantly, compromised for certain molybdenites.
The Decoupling Enigma
Early in the development of molybdenite geochronology it was empirically established that some Archean molybdenites yielded different Re-Os ages for different size fractions of mechanically separated grains. This was the first indication that the daughter 187Os may migrate within a molybdenite crystal, as Os is not a good crystallographic fit (hence, no common Os in molybdenite). The first LA-ICP-MS investigation of this phenomenon yielded both >3 Ga and future spot ages (Stein et al. 2003). Osmium was clearly migrating and taking residence in dislocation features and delamination cracks. Decoupling was linked to older molybdenites with orogenic overprints. Based on additional LA-ICP-MS work, it was advocated that 40 mg aliquants from a large mineral separate were necessary to overcome decoupling in older coarse-grained molybdenite (Selby and Creaser 2004).
Download high-resolution image
However, it is now widely agreed that very small samples (sub-mg) are just as capable of yielding robust ages as larger samples. What matters is context, not milligrams, and taking the whole molybdenite crystal from its silicate matrix (Stein 2006). Crystal fragments or thin sheets of molybdenite from (older) samples with coarse-grained molybdenite may not yield accurate ages.
Model Versus Isochron Ages
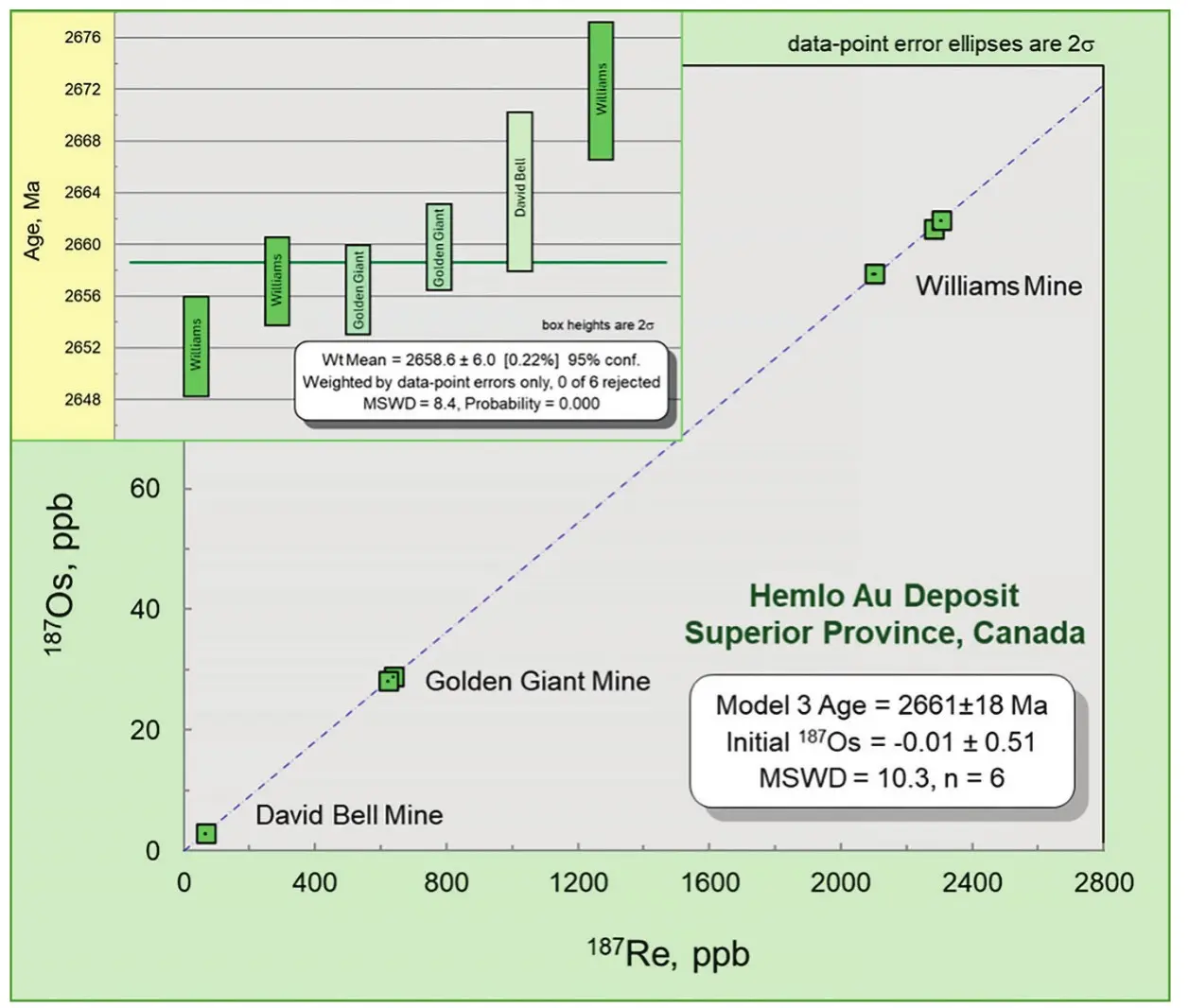
Despite the power of Re-Os molybdenite dating, interpretation of Au-associated molybdenite ages can be complex, especially in multiply metamorphosed and structurally complex terranes. Figure 6 (inset) shows Re-Os model ages for three mines in the Hemlo district versus an isochron age for the same Re-Os data. The Archean terrane hosting Hemlo contains multiple metamorphic overprints and visibly deformed molybdenite. This setting presented an early testing ground for molybdenite’s response to severe overprinting. When acquired in 2002, these data were open to an interpretation of isotopic disturbance, as Hemlo resides in a major sinistral shear zone in a sedimentary basin with intrusions and an amphibolite-grade metamorphic overprint. More than 20 years later and having dated many varieties of Au deposits, we now know that the more plausible explanation for small age differences is a pulsed history of Au-formation. For Re-Os geochronology, this was an early lesson that less precise isochrons with notably high MSWDs are telling a story and, for molybdenites in complex terranes, individual Re-Os model ages should be reported rather than a weighted mean age.
THE MOVE TO ARSENOPYRITE AND PYRITE
The surprising discovery that arsenopyrite and pyrite can have molybdenite-like Os isotopic compositions came from the Bendigo Au deposit (Victoria, Australia), where an arsenopyrite-pyrite isochron with a well-defined crustal Os initial ratio provided an accurate age for Au mineralization (Arne et al. 2001).
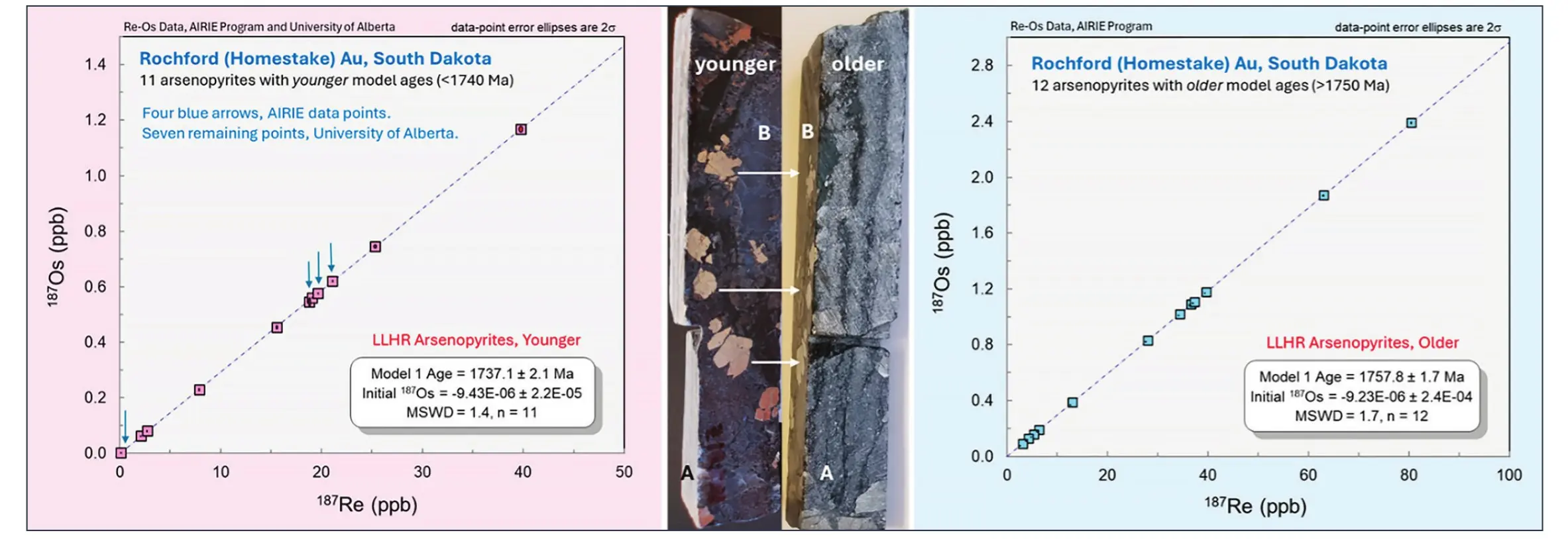
Download high-resolution image
Arsenopyrite and pyrite are widely associated with many styles of Au mineralization, whereas molybdenite is rare to absent. Arsenopyrite became a mineral of choice for Re-Os dating, as explored in several papers (e.g., Morelli et al. 2005). In a study based on a single sample, Morelli et al. (2010) dated presumed co-genetic arsenopyrite and pyrrhotite from the renowned Homestake Au mine in South Dakota to “pinpoint the timing of Au mineralization.” A precise arsenopyrite isochron resulted, whereas the presumed co-genetic pyrrhotite data were highly scattered. Given the amphibolite grade host rocks for the Homestake Au deposit, Morelli et al. (2010) concluded that the Re-Os chronometer is robust to at least 400 °C for arsenopyrite, while pyrrhotite is disturbed at 300–350 °C. In Figure 7, their Homestake data are combined with Rochford-Homestake data (AIRIE) to illustrate the potential limitations of looking at a single sample from a complex Au-bearing terrane. Taken together, results from the two labs provide a powerful analysis of multi-step processes in forming a major Au endowment.
Pyrite on its own has been successfully used for Re-Os geochronology in a variety of geologic settings. As one of the most common sulfide minerals in hydrothermal ores, the ability to date pyrite has utility for almost all ore deposit types. Like arsenopyrite, Re and Os abundances in pyrite are typically ppb to ppt level. High-precision Re-Os pyrite ages have been determined from carbonate-hosted Pb-Zn (Irish type and Mississippi Valley type), sedimentary exhalative (SEDEX-type), volcanogenic massive sulfides (VMS), orogenic gold, iron oxide Cu-Au (IOCG), and other deposit types (see Rooney reference in Toolkit). The first landmark paper analyzing pyrite (Stein et al. 2000) was followed by detailed studies producing Re-Os pyrite isochron ages, including some with <1% two-sigma uncertainty (Alberta laboratory) that decisively tested competing models of ore formation. Pyrite geochronology has been used in metamorphic terrane studies yielding robust Re-Os ages at ~660 °C (van Acken et al. 2014). Rhenium incorporation into pyrite appears to involve both lattice-bound Re as well as Re-rich micron-scale inclusions (Hnatyshin et al. 2020).
Arsenopyrite, pyrrhotite, and pyrite studies have revealed temperature limits for the stability of the Re-Os chronometer in particular cases. However, no two ore-forming environments are alike and the role of chemistry, especially oxidation, may be underestimated. For molybdenite, it appears that oxidation, not temperature, decides the fate of its radiometric clock (see Stein references in Toolkit), which remains stable through very high-grade metamorphism (>900 °C).
Sky’s the Limit
From pyrite and arsenopyrite, the sky opened, and any sulfide could now be considered for Re-Os dating. However, except for molybdenite, sulfides should not be labeled as “good for dating” or “not good for dating.”

Download high-resolution image
In fact, many arsenopyrites and pyrites do not deliver good age results for many different reasons. What matters is (1) the sample’s ability to include and retain Re-Os, (2) potential postdepositional exposure to oxidation (which is fatal for Re-Os dating), and (3) the strategy for extraction of the material to be dated. Meaningful geochronology also requires understanding of the local and wider geologic context of the samples, which is true for any geochronometer.
The Re-Os geochronological potential of many other sulfide minerals has been explored, including the Cu and/or Co minerals bornite, chalcopyrite, and cobaltite, as well as arsenides and sulfarsenides such as safflorite, gersdorrfite, and rammelsbergite (see Rooney reference in Toolkit). Many of these are critical minerals containing Co and Ni, needed in current battery technology for the energy transition towards decarbonation. This is also true of both metamorphic and hydrothermal graphite, whose Re-Os systematics have been investigated recently (see Toma reference in Toolkit). Also very recently, Re-Os has been used to define and correlate episodes of supergene (metal) enrichment in Cu porphyry deposits (AIRIE). As was the case for molybdenite, such studies are opening a new era of resource geoscience research that is already finding new applications in the broader geosciences (Fig. 8).
WHAT LIES AHEAD?
The historical context presented in this review is just the beginning. The Re-Os isotope system holds a critical position in resource geosciences, providing direct informa- tion on the timing of ore formation in base metal systems (Cu, Pb, Zn), precious metal systems (Au, Ag), and critical metal systems (Co, Ni, C). It was born out of the meteorite and mantle community and step-by-step has climbed a ladder that could not have been imagined 25 years ago (Fig. 8).
The future will certainly see ever broadening applications. For example, Re-Os dating of pyrite to date the rise of oxygen in Earth’s early atmosphere (see Hannah reference in Toolkit) has received substantial interest from planetary scientists and geobiologists. The ability to measure increas- ingly smaller concentrations of Os will allow the medical community to expand its investigation of Os toxicity, as volatile oxygenated Os species (OsO₄, HOsO₅⁻, H₃OsO₆⁻) are potential health risks in communities anchored in Os-rich ultramafic bedrock and their related groundwater sources. Studies that pair Os-Hg may provide new insights into extinction records. More generally, the combined use of multiple isotopic tracers vastly improves our ability to develop and test plausible hypotheses. The Re-Os isotope system remains underutilized in this regard.
The chemistry and geology communities have much to learn from one another. While chemists construct perfect crystals and functional MOFs (metal-organic frameworks), what nature has created with all its warts and heteroge- neities may give chemists new insights. The hydrocarbon and economic geology communities could work together more effectively, as metals and hydrocarbons are intimately associated (Fig. 9). For years, these resources have been studied as separate subjects, but the equation is simple. Kerogen sequesters metals. Maturation of kerogen to generate hydrocarbons is a first step in releasing metals. Migration of hydrocarbons transports metals, poten- tially adding a previously unrecognized contaminant to groundwater.
Re-Os will find increasing application in legal circles. Metal contamination derived from mining operations, present and past, may be fingerprinted (forensics) through its Os isotopic composition.

Download high-resolution image
As rivers collect waters from many upstream tributaries, Os isotopes can be used to identify the headwaters draining a contaminant source, thereby assigning responsibility (blame) to the correct party (more forensics), or alternatively indicating an unusually high, but natural, background level in local soils and bedrock (mother nature is to blame).
Rhenium-osmium molybdenite dating by LA-ICP-MS/ MS has been tested by several working groups and most recently has led to characterization of two samples for use as LA-ICP-MS/MS reference materials (Tamblyn et al. 2024). At present, the technique is limited to geologically old molybdenites with relatively high Re. The advantage of laser ablation is the rapid acquisition of large numbers of micron-scale spot ages (to be averaged) and the poten- tial recognition of textural domains and growth zones. Therein lies the complication of scale (Zimmerman et al. 2022). Individual spot ages may be disparate, imprecise, and even geologically non-sensical, but when pooled, may provide meaningful ages matching robust, high-precision milligram-scale ID-NTIMS ages. New reference materials require systematically designed proof of reproducibility using LA-ICP-MS/MS and their ages must be validated against systematically reproduced ID-NTIMS ages. The statistical rigor required by NIST to market the Henderson molybdenite Re-Os reference material for its age reproduc- ibility (Markey et al. 2007) provides a template for develop- ment of a reference material for LA-ICP-MS/MS analyses.
CLOSING REMARKS
The historical context presented in this review and throughout this Elements issue highlights some of the early landmark papers and the challenges faced by several teams working to bring the Re-Os chronometer into the crustal realm. Different teams used their different backgrounds to meet the new opportunities offered by the Re-Os tracer and clock. The open cooperation in this early research included sharing information, opening laboratories to one another for visits, writing letters of support for one another, and regularly catching up at meetings to learn from one another. It was not perfect, and occasionally there were surprises and twinges of jealousy, but sharing the excite- ment brings out the best in everyone and moves science forward effectively.
ACKNOWLEDGMENTS
HS is grateful to John W. Morgan for his mentorship, and to Du Andao for her friendship. After the USGS terminated our nascent Re-Os work in 1996, I am forever thankful that Richard Markey, my USGS research associate, took the chance and joined me in Colorado to build AIRIE. After 12 years, Rich took up teaching science in high school, but without him in the early years, I would have had a different career. I am deeply grateful to Co-Editor Laurie Reisberg, who significantly improved this chapter on short notice, for her unwavering friendship and her nightly email sign-off of “bon courage.” This issue is the last for our Principal Editor, Janne Blichert-Toft. When I approached Janne about an Elements issue on Re-Os she was immediately enthusiastic and brilliantly wrapped in Laurie Reisberg as Co-Editor. Janne’s keen insight and red pen will be missed. Executive Editor Esther Posner has been wonderful and patient; she operates in a spirit of calm. Together, we made this journey, keeping one another in line, and in (mostly) good spirits given the challenges of recent times. I am grateful to my co-authors and the many others who have been part of the molybdenite experience. Special thanks go to three reviewers, at three different career levels, Randall Karcher, Douglas Kreiner, and Judy Hannah for thoughtful comments.
REFERENCES
Arne DC, Bierlein FP, Morgan JW, Stein HJ (2001) Re-Os dating of sulfides associated with gold mineralization in central Victoria, Australia. Economic Geology 96: 1455-1459, doi: 10.2113/ gsecongeo.96.6.1455
Bingen B, Stein H (2003) Molybdenite Re–Os dating of biotite dehydration melting in the Rogaland high-temperature granulites, S Norway. Earth and Planetary Science Letters 208: 181-195, doi: 10.1016/ S0012-821X(03)00036-0
Cannell J (2004) El Teniente porphyry copper-molybdenum deposit, central Chile. PhD thesis, University of Tasmania, 317 pp
Higgins MD, Smith DGW (2010) A census of mineral species in 2010. Elements 6: 346
Hirt B, Herr W, Hoffmeister W (1963) Age determination by the rheniumosmium method. In: Radioactive Dating. International Atomic Energy Agency, Vienna, pp 35-43
Hnatyshin D and 5 coauthors (2020) Understanding the microscale spatial distribution and mineralogical residency of Re in pyrite: examples from carbonatehosted Zn-Pb ores and implications for pyrite Re-Os geochronology. Chemical Geology 533: 119427, doi: 10.1016/j. chemgeo.2019.119427
Luck JM, Allègre CJ (1982) The study of molybdenites through the 187Re–187Os chronometer. Earth and Planetary Science Letters 61: 291-296, doi: 10.1016/0012-821X(82)90060-7
Maksaev V and 6 coauthors (2004) New chronology for El Teniente, Chilian Andes, from U-Pb, 40Ar/39Ar, Re-Os, and fissiontrack dating: implications for the evolution of a supergiant porphyry Cu-Mo deposit. In: Sillitoe RH, Perelló J, Vidal CE (eds) Andean Metallogeny: New Discoveries, Concepts, and Updates. Society of Economic Geologists Special Publication 11: 15-54, doi: 10.5382/SP.11.02
Markey R, Stein H, Morgan J (1998) Highly precise Re–Os dating of molybdenite using alkaline fusion and NTIMS. Talanta 45: 935-946, doi: 10.1016/ S0039-9140(97)00198-7
Markey R, Hannah JL, Morgan JW, Stein HJ (2003) A double spike for osmium analysis of highly radiogenic samples. Chemical Geology 200: 395-406, doi: doi. org/10.1016/S0009-2541(03)00197-9
Markey R and 5 coauthors (2007) Standardizing Re-Os geochronology: a new molybdenite reference material (Henderson, USA) and the stoichiometry of Os salts. Chemical Geology 244: 74-87, doi: doi.org/10.1016/j.chemgeo.2007.06.002
McCandless TE, Ruiz J, Campbell AR (1993) Rhenium behavior in molybdenite in hypogene and near-surface environments: implications for Re-Os geochronometry. Geochimica et Cosmochimica Acta 57: 889-905, doi: 10.1016/0016-7037(93)90176-w
Morelli RM, Creaser RA, Selby D, Kontak DJ, Horne RJ (2005) Rhenium-osmium geochronology of arsenopyrite in Meguma Group gold deposits, Meguma Terrane, Nova Scotia, Canada: evidence for multiple gold-mineralizing events. Economic Geology 100: 1229-1242, doi: 10.361-0128/05/3542/1229-14
Morelli RM, Bell CC, Creaser RA, Simonetti A (2010) Constraints on the genesis of gold mineralization at the Homestake gold deposit, Black Hills, South Dakota from rhenium–osmium sulfide geochronology. Mineralium Deposita 45: 461-480, doi: 10.1007/s00126-010-0284-9
Raith JG, Stein HJ (2000) Re–Os dating and sulfur isotope composition of molybdenite from tungsten deposits in western Namaqualand, South Africa: implications for ore genesis and the timing of metamorphism. Mineralium Deposita 35: 741-753, doi: 10.1007/s001260050276
Selby D, Creaser RA (2004) Macroscale NTIMS and microscale LA-MC-ICP-MS Re-Os isotopic analysis of molybdenite: testing spatial restrictions for reliable Re-Os age determinations, and implications for the decoupling of Re and Os within molybdenite. Geochimica et Cosmochimica Acta 68: 3897-3908, doi: 10.1016/j.gca.2004.03.022
Selby D, Creaser RA, Feeley M (2004) Accurate and precise Re-Os molybdenite dates from the Galway Granite, Ireland: critical comment to “Disturbance of the Re-Os chronometer of molybdenites from the late-Caledonian Galway Granite, Ireland, by hydrothermal fluid circulation” by Suzuki et al. Geochemical Journal, 35, 29-35, 2001. Geochemical Journal 38: 291-294, doi: 10.2343/geochemj.38.291
Selby D, Creaser RA, Stein HJ, Markey RJ, Hannah JL (2007) Assessment of the 187Redecay constant by cross calibration of Re–Os molybdenite and U–Pb zircon chronometers in magmatic ore systems. Geochimica et Cosmochimica Acta 71: 1999-2013, doi: 10.1016/j.gca.2007.01.008
Stein HJ, Morgan JW, Scherstén A (2000) Re-Os dating of low-level highly radiogenic (LLHR) sulfides: the Harnäs gold deposit, southwest Sweden, records continental- scale tectonic events. Economic Geology 95: 1657-1671, doi: 10.2113/ gsecongeo.95.8.1657
Stein HJ, Bingen B (2002) 1.05–1.01 Ga Sveconorwegian metamorphism and deformation of the supracrustal sequence at Sæsvatn, south Norway: Re-Os dating of Cu-Mo mineral occurrences. In: Blundell D, Neubauer F, von Quadt A (eds) The Timing and Location of Major Ore Deposits in an Evolving Orogen. Geological Society, London, Special Publications 204, pp 319-335, doi: 10.1144/GSL. SP.2002.204.01.18
Stein H, Scherstén A, Hannah J, Markey R (2003) Subgrain-scale decoupling of Re and 187Os and assessment of laser ablation ICP-MS spot dating in molybdenite. Geochimica et Cosmochimica Acta 67: 3673-3686, doi: 10.1016/ S0016-7037(03)00269-2
Stein HJ (2006) Low-rhenium molybdenite by metamorphism in northern Sweden: recognition, genesis, and global implications. Lithos 87: 300-327, doi: 10.1016/j. lithos.2005.06.014
Suzuki K, Shimizu H, Masuda A (1996) Re–Os dating of molybdenites from ore deposits in Japan: implication for closure temperature of the Re–Os system for molybdenite and the cooling history of molybdenum ore deposits. Geochimica et Cosmochimica Acta 60: 3151-3159, doi: 10.1016/0016-7037(96)00164-0
Suzuki K, Feely M, O’Reilly CO (2001) Disturbance of the Re-Os chronometer of molybdenites from the late-Caledonian Galway Granite, Ireland, by hydrothermal fluid circulation. Geochemical Journal 35: 29-35, doi: 10.2343/geochemj.35.29
Tamblyn R and 8 coauthors (2024) Molybdenite reference materials for in situ LA-ICP-MS/MS Re-Os geochronology. Geostandards and Geoanalytical Research 48: 393-410, doi: 10.1111/ggr.12550
van Acken D, Su W, Gao J, Creaser RA (2014) Preservation of Re-Os isotope signatures in pyrite throughout low-T, high-P eclogite facies metamorphism. Terra Nova 26: 402-407, 10.1111/ter.12113
Watanabe Y, Stein HJ, (2000) Re-Os ages for the Erdenet and Tsagaan Suvarga porphyry Cu-Mo deposits, Mongolia, and tectonic implications. Economic Geology 95: 1537-1542, doi: 10.2113/ gsecongeo.95.7.1537
Yang G, Zimmerman A, Stein H, Hannah J (2015) Pretreatment of nitric acid with hydrogen peroxide reduces total procedural Os blank to femtogram levels. Analytical Chemistry 87: 7017-7021, doi: 10.1021/acs.analchem.5b01751
Zimmerman A, Stein HJ, Morgan JW, Markey RJ, Watanabe Y (2014) Re–Os geochronology of the El Salvador porphyry Cu-Mo deposit, Chile: tracking analytical improvements in accuracy and precision over the past decade. Geochimica et Cosmochimica Acta 131: 13-32, doi: 10.1016/j.gca.2014.01.016
Zimmerman A, Yang G, Stein HJ, Hannah JL (2022) A critical review of molybdenite 187Reparent-187Os daughter intra-crystalline decoupling in light of recent in situ micro-scale observations. Geostandards and Geoanalytical Research 46: 761-772, doi: 10.1111/GGR.12448

